
|

|
|
Transplant Statistics: Annual Report : Kidney and Pancreas Transplantation
This chapter reviews data regarding kidney and pancreas transplantation in the United States from 1992 through 2001, providing a unique summary of the evolution of these fields over the past decade[Tables 5.1-5.10]. The first section examines kidney transplantation, with subsections addressing the waiting list, characteristics of transplant recipients, patient survival and death rates after transplantation, and graft survival. Following the kidney data are sections addressing simultaneous kidney-pancreas transplantation (SPK)[Tables 8.1-8.10] and isolated pancreas transplantation — both pancreas transplant alone (PTA)[Tables 6.1-6.10] and pancreas after kidney (PAK)[Tables 7.1-7.10].
During the past decade, kidney transplantation has increasingly been recognized as the treatment of choice for medically suitable patients with End Stage Renal Disease (ESRD) (1). Medicare statutes now mandate that each ESRD patient undergo evaluation regarding candidacy for transplantation, and referrals to transplant centers have increased dramatically (2). Transplants from deceased donors rose from 7,202 in 1992 to 8,202 in 2001, an increase of 14% [Table 5.4a]. In contrast, the number of transplants from living donors more than doubled, from 2,535 in 1992 to 5,969 in 2001 (a 135% increase)[Table 5.4b]. At the same time, a dwindling list of absolute and relative contraindications for kidney transplantation has changed the profile of the average transplant candidate, who is now more likely to be older and burdened with greater comorbidity than previously thought acceptable (3,4).
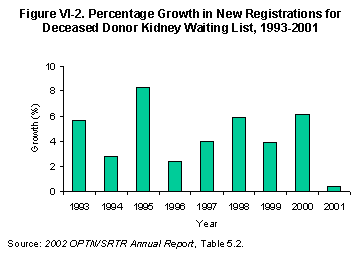
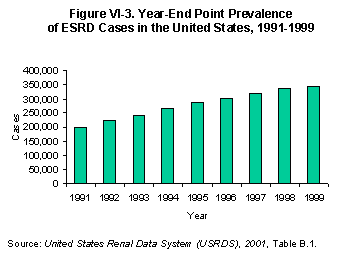
The waiting list for kidneys from deceased donors expanded dramatically over the past decade. At the end of 2001, there were 51,144 candidates awaiting transplantation, compared to 22,063 in 1992, an increase of 132% (Figure VI-1)[Table 5.1]. However, the rate of growth slowed substantially in 2001 (Figure VI-2). This reduced pace coincides with the recent slower growth in year-end point prevalence of ESRD (Figure VI-3) (5).
This slowdown may also be due, in part, to increases in living donor transplants, reflecting both application of newer technologies (e.g., laparoscopic donor nephrectomy) and changing eligibility criteria that allow almost any healthy, ABO-compatible adult to be a kidney donor (6,7).
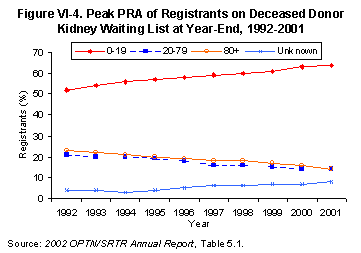
There was little change in the relative percentage of men and women on the waiting list during the past decade, or in the relative proportions of ABO blood types among recipients[Table 5.1]. The number of registrants reporting a prior organ transplant declined relative to the number of registrants awaiting a primary transplant. The proportion of sensitized patients, as measured by peak panel reactive antibody (PRA) =20%, continued to decline (Figure VI-4)[Table 5.1]. Increased erythropoietin use and the consequent lack of sensitizing blood transfusions, along with fewer early graft losses requiring retransplantation, may explain this observation (8). Women and African Americans were more likely to be sensitized (PRA =20%) than men and members of other racial and ethnic groups.
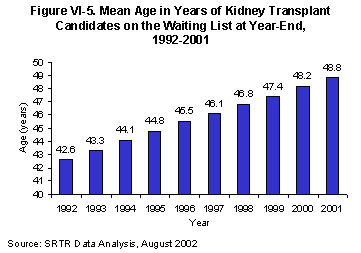
The mean age of the candidates on the kidney transplant waiting list continued to rise over the decade (rising from 42.6 in 1992 to 48.8 in 2001). Similarly, the percentages of the waiting list made up of individuals aged 50-64 and 65 or older continued to grow (from 31% in 1992 to 51% in 2001) [Table 5.1]. (Table VI-1, Figure VI-5 and VI-6). Absolute increases in the number of candidates waiting were observed for all age groups, with the exception of infants younger than 1 year old[Table 5.1]. United States Renal Data System year-end point prevalence counts by age revealed trends that were similar, though less dramatic (5). The increasing age of those awaiting deceased donor kidney transplantation likely reflects liberalized criteria for listing, as well as corresponding changes in the ESRD population.
Table VI-1. Percentages of Candidates Over 65 Years Old on the Kidney Waiting List, and Among Recipients of Deceased Donor and Living Donor Transplants
| Year | Waiting List* | Deceased Donor Transplants** | Living Donor Transplants** |
|---|---|---|---|
| 1992 | 4.9% | 5.1% | 2.1% |
| 2001 | 12.1% | 10.9% | 6.3% |
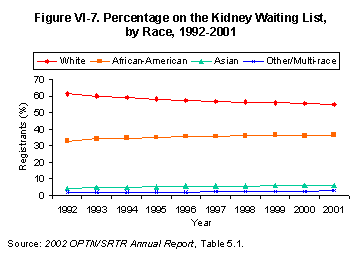
The proportion of racial and ethnic minorities on the waiting list continued to slowly but steadily grow (Figure VI-7)[Table 5.1]. The proportion of Hispanic/Latino candidates experienced a similar small increase, though the trend for non-Hispanic/non-Latino candidates was obscured by changes in reporting practice. Overall, at year-end 2001, nonwhite candidates comprised 45% of the waiting list; this figure stood at 44% in 2000 and 38% in 1992.
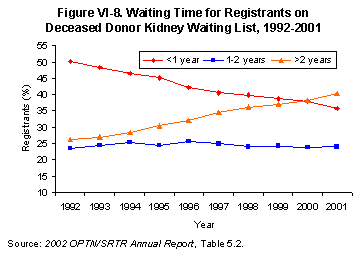
Time spent on the waiting list continues to increase. The proportion of registrants waiting longer than two years reached 40% in 2001, up from 26% in 1992 (Figure VI-8) [Table 5.1]. The combination of increasing age and waiting time among those listed for transplant has given rise to a series of new clinical initiatives. Known collectively as “waiting list management,” these initiatives have grown out of transplant centers’ efforts to ensure that those awaiting transplantation remain medically suitable for transplantation (9).
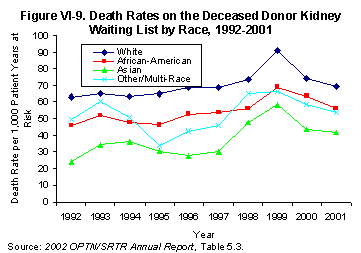
Reported deaths on the waiting list and annual death rates (per 1,000 patient years at risk) showed a modest increase over the past 10 years. In 1992, the annual death rate was 56 and in 2001 it was 63, having risen as high as 80 in 1999[Table 5.3]. However, death rates were not uniform for all classes of candidates. Death rates for candidates 50 to 64 years of age showed little change between 1992 and 2001, (72 and 76, respectively) and declined by 10% (48 to 43) for patients aged 18-49[Table 5.3]. Whites have a higher death rate on the waiting list than other racial groups at all time points examined. In 2001, the death rate for whites was 70, compared with 56, and 42, for African Americans, and Asians, respectively (Figure VI-9). Death rates for Hispanics/Latinos were 51 compared to 64 for non-Hispanics/non-Latinos. Finally, as might be predicted, gender and ABO grouping do not appear to influence death rates on the waiting list. With a waiting list more heavily populated by older and minority patients in 2001 than in 1992, the overall change in death rates is difficult to interpret, as are the noted racial and ethnic discrepancies[Table 5.3]. Indeed, the demographics of death on the waiting list mimic the demographics of death among dialysis patients.
The ideal outcome for wait-listed ESRD patients is to receive a kidney in a timely fashion. Unfortunately for those awaiting kidneys, deceased donor transplantation was less likely in 2001 than in 1992. Overall, in 1992, a registrant had a 33% chance of receiving a kidney from a deceased donor within a given year. In 2001, the chance had declined to 16%. There are wide disparities regarding the likelihood of transplantation by level of presensitization and blood group. Over 20% of wait-listed unsensitized patients receive kidneys each year; however, a peak PRA =20% reduces those chances to 13% or less, depending on degree of sensitization. Regarding ABO blood type, 33% of blood type AB patients can expect to be transplanted within a year, as can 22% of ABO type A candidates. In contrast, patients with ABO types B or O have transplantation rates of 11%-13%[Tables 5.1 and 5.4a].
Age and ethnicity also affect the likelihood of transplantation. In recognition of the adverse effects of renal insufficiency on growth and development, patients younger than 18 are awarded sufficient additional points to generate an overall annual transplantation rate of 41% (Table VI-2). The allocation algorithm also awards points for HLA similarity. Thus, those candidates easiest to match (whites, who bear the greatest genetic similarities to the donor population) have a higher annual rate of transplantation (19%) when compared to other racial groups (12%-14%). These findings are consistent with a recent SRTR study prepared for the OPTN/UNOS Minority Affairs Committee that documented these same trends in multivariate analyses (10). While previously transplanted recipients accounted for a relatively stable 13% of deceased donor transplants, the chances of a wait-listed patient being retransplanted fell from nearly 24% in 1992 to 11% in 2001 [Tables 5.1 and 5.4a]. These trends directly reflect choices made in the allocation process, which rewards HLA matching, youth, and time on the waiting list.
Table VI-2. Current OPTN/UNOS Point System for Allocation of Cadaveric Kidneys
| Criterion | Points Awarded |
|---|---|
| HLA Mismatches | |
| No A, B, or DR mismatches | Mandatory sharing |
| No B or DR mismatches | 7 |
| 1 B or DR mismatch | 5 |
| Total of 2 mismatches at B and DR loci | 2 |
| Presensitization | |
| PRA >= 80% | 4 |
| Waiting time | |
| Longest wait on OPO list | 1 |
| Shorter waits | Fractions of 1 |
| Each year on list | 1 |
| Age | |
| < 11 years | 4 |
| 11-18 years | 3 |
Accordingly, waiting times to transplantation have increased. For example, the proportion of candidates who had waited for 6 to 12 months for a deceased donor transplant decreased from 21% in 1992 to 17% in 2001, and those who had been waiting for 3 to 5 years increased from 9% in 1992 to 16% in 2001[Table 5.1]. The same variables that influence likelihood of transplantation impact time from wait-listing to transplantation[Table 5.2]. Whites added to the list in 1992 waited a median time of 511 days for a suitable kidney, and African Americans had a median waiting time of 970 days. For patients listed in 1997 (the most recent listing date for which such calculations can be performed), time to transplant lengthened dramatically, to 675 days (up 32%) for whites and 1,603 days (up 65%) for African Americans. Other minority patients have experienced lengthening waiting times during the past decade with waits that are longer than those observed for whites, but not as long as those seen in African Americans, with the same trend for longer waits over time (Figure VI-10). In general, these differences are documented not only in national data, but confirmed when analyzed in individual regions or OPO service areas (10,11).
Transplant candidates with ABO blood types B and O wait longer than those with A or AB blood types, with even greater disparity in recent years. (Figure VI-11) A regional analysis confirms this trend. Among the cohort of patients wait-listed in 1998, in 10 of 11 OPTN/UNOS regions candidates with blood type B had the longest wait before the first quartile of candidates received transplants; those with blood type O had the second longest wait. (In the remaining region, type O candidates had the longest waiting time and type B candidates had the second longest.) Similarly, in 8 of 11 regions, type B candidates had the longest and type O the second longest median waiting times to transplantation. In another region, type O candidates had the longest waiting time and type B candidates had the second longest; in the remaining two regions, waiting times for candidates with blood types O and B were so long that median intervals to transplantation could not be calculated.
Blood type B is more common among African Americans than it is among whites. As would be expected, a similar pattern was seen when waiting times for the cohort wait-listed in 1998 were analyzed by race. In 10 of the 11 OPTN/UNOS regions, African Americans experienced the longest time to transplant among the first quartile of wait-listed candidates; Asians had the longest wait in the 11th. African Americans also had the longest median waiting times in all 8 regions for which median waiting times could be determined.
The difference in waiting times between blood type A and B candidates is the subject of a new UNOS voluntary variance, adopted in 2001, which allows transplantation of type A2 and A2B deceased donor kidneys into type B candidates. This action is expected to increase transplantation rates for blood type B candidates. Since type B is found more often in African Americans than in whites, it is also hoped that this new variance will decrease waiting times for minorities (12).
Sensitized patients (PRA =20%) wait over twice as long as unsensitized candidates. For highly sensitized patients (PRA =80%) the time required for 25% of candidates to receive a cadaveric kidney transplant is about the same duration in recent years as in 1992; waiting times were too long to calculate the median time to transplantation. Women, on the whole, experience somewhat longer waiting times than men; however, this difference is not observed among unsensitized (0%-19% PRA) men and women[Table 5.2]. These findings may be attributable to the sensitization that occurs in women as a result of exposure to paternal antigens during pregnancy.
Characteristics of those patients receiving transplants from deceased donors have changed during the last decade, in large part mirroring changes in the waiting list. The proportion of transplants performed in young people (under 35 years old) declined from 29% in 1992 to 19% in 2001[Table 5.4a]. Conversely, the percentage of deceased donor kidneys going to older recipients increased, rising for both 50-64 (up from 28% in 1992 to 38% in 2001) and for those 65 and older (up from 5% in 1992 to 11% in 2001). The proportion of whites receiving transplants decreased from 70% in 1992 to 64% in 2001, and the proportion of African American recipients rose from 24% to 30% during the same period. The proportion of Asian recipients remained about the same. Similarly, over the past decade, the proportion of Hispanic/Latino recipients increased modestly.
As discussed above, HLA matching, as defined in the OPTN/UNOS allocation algorithm, continues to exert significant influence over the distribution and timing of deceased donor kidney transplantation. The proportion of deceased donor kidneys transplanted with no HLA-A, B, or DR mismatches has increased over the past 10 years, from 7% in 1992 to 15% in 2001[Table 5.4a]. The biggest increase during this period occurred between 1994 and 1995. During this time, the definition of optimally-matched kidneys, which are mandatorily shared and previously limited to phenotypic identity between donor and recipient, was broadened to include transplantation between zero-antigen mismatched donor and recipient pairs (13). For patients transplanted between 1995 and 2001, SRTR analysis shows recipients of zero-mismatched kidneys experienced the shortest median time to transplantation (9.7 months). There has been a more gradual increase in the proportion of kidneys transplanted with five or six mismatches, from 21% in 1992 to 29% in 2001. Patients who receive these kidneys tend to have waited longest for a transplant of all HLA matching groups (18.7 months). These trends indicate that an ESRD patient likely to receive a well-matched kidney typically receives that organ soon after wait-listing. Conversely, those patients difficult to match tend to spend more time on the list, and ultimately receive a poorly-matched kidney.
Deceased donor transplantation is still the mainstay of kidney transplantation in the United States; the majority of functioning renal allografts originated from deceased donors (5). The first year in which living kidney donors outnumbered deceased kidney donors (5,974 vs. 5,528) in the United States [Table 1.1] was 2001. However, because most deceased donors generate more than one kidney, this source still accounted for 57% of kidney transplants performed in 2001 (Figure VI-12)[Table 1.8]. Nonetheless, rapid growth in the numbers of living donor kidney recipients in recent years indicates the increasing importance of living donors in making transplantation available to more ESRD patients.
From 1992 to 2001, the percentages of ESRD patients older than 65 increased for candidates on the waiting list, recipients of cadaveric kidneys, and recipients of living donor kidneys (Table VI-1). Recipients older than 50 made up 18% of living donor transplants in 1992 and 36% in 2001[Table 5.4b]. In absolute numbers, living donor kidney transplants in this age group rose from 446 to 2,131, or 378%. This contrasts with the relatively unchanged number of children receiving living donor transplants, and growth for living donor kidney recipients aged 18-49 that paralleled the growth in the waiting list. Whites continued to receive the overwhelming majority of kidneys from living donors (78%), with minority patients’ percentages largely unchanged over the last decade[Table 5.4b].Schweitzer and colleagues suggest that at least part of this ethnic differential might be overcome with more comprehensive educational efforts (14). HLA matching in living donor transplantation also underwent a dramatic shift since 1992, with the fraction of living donor transplants going to two-haplotype matched recipients declining from 20% to 11% in 2001. At the same time, the percentage of living donor recipients with greater than one-haplotype mismatch has increased from 10% to 31%. Presumably, this change represents increasing utilization of spouses and other unrelated donors (15).
Recognizing the increasing importance of the living donor, the SRTR recently performed an analysis to determine the likelihood of various patient groups receiving kidneys from living donors or deceased donors. This analysis included 49,128 kidney transplants (79% from deceased donors) performed between March 6, 1995 and December 31, 2000. Table VI-3 contains the demographic data derived from this large sample of kidney recipients.
Younger patients and whites received a greater proportion of living donor kidneys than older patients and minorities. Recipients of living donor kidneys were, in general, more likely to be better educated, employed, to live in counties with higher average family income, to have private health insurance, and to reside in OPO areas with longer median waiting times. They also spent less time on the waiting list, with mean waiting time to transplantation nearly twice as long for deceased donor kidney recipients (563 days) as for living donor kidney recipients (292 days). This discrepancy is likely even wider than the numbers suggest, as a substantial proportion of living donor recipients are never placed on the waiting list. These waiting times assume greater significance in light of recent data indicating that the best outcomes after transplantation accrue to those patients transplanted early in the course of ESRD (16).
Table VI-4 provides another view of this same set of recipients and indicates that, in terms of absolute numbers, deceased donor kidney recipients dominate most every clinical measure. Even for the youngest recipients, transplantation from deceased donors was more common. Fewer older recipients received kidneys from living donors as well: only 13% of those over age 65. While males are generally more likely to be transplanted, Table VI-4 shows that a slightly higher percentage of females who were transplanted received a living donor kidney (22% versus 21% for males). Several patient characteristics were associated with higher percentages receiving a living donor transplant than the 21% overall average. These include higher education, better insurance, and being employed.
Considerable work over the past several years has conclusively established the life-saving nature of renal transplantation (1,17,18). Recipient age, race, and underlying renal disease affect patient survival both on the waiting list and following kidney transplantation. Proposals to incorporate variables affecting patient survival in kidney allocation algorithms have been made (19). Regardless, these remain important considerations in counseling patients on their suitability for kidney transplantation.
The overall annual posttransplant death rate, calculated as deaths per 1,000 patient years at risk, remained remarkably stable over the past decade, ranging from 42 to 54[Table 5.7]. In contrast, as noted above, the death rate on the waiting list appeared to be increasing through 1999 [Table 5.3]. It is notable that the waiting list death rate consistently exceeds that observed following transplantation, mirroring death rates for dialysis and transplanted patients in general (1,5).
Older patients now constitute much larger proportions of those on the waiting list and receiving transplants than they did 10 years ago. As would be expected, death rates increase progressively with age, both for patients on the transplant waiting list [Table 5.3] and for those who have undergone deceased donor or living donor renal transplantation[Table 5.7].
Encouragingly, posttransplant death rates have decreased over the past decade for recipients aged 50-64 and have remained steady for those 65 and over[Table 5.7]. Five-year survival for recipients 65 and over (deceased donor kidney), however, is fully 30% less than for recipients aged 18-34 years (58% vs. 90%). Differences in five-year survival by age after living donor transplantation, while still evident, are substantially less than for deceased donor kidneys (Figure VI-13)[Table 5.9b]. Among younger recipients, children less than 6 years of age have high death rates, both on the waiting list[Table 5.3] and following transplantation[Table 5.7].
Table VI-3. Distribution of Recipient Characteristics Among Living vs. Deceased Donor Kidney Transplants, 1995-2000 (Column Percent)
| Measure | Living | Deceased | Measure | Living | Deceased | ||
|---|---|---|---|---|---|---|---|
| Transplants | Education (%) | ||||||
| Sample size (n) | 10,331 | 38,797 | None | 0.4 | 0.7 | ||
| Percent | 21.0 | 79.0 | 0-8 years | 5.0 | 6.4 | ||
| Age (%) | High school grad | 32.3 | 33.8 | ||||
| 0-9 | 1.1 | 1.2 | College | 32.7 | 24.1 | ||
| 10-17 | 3.0 | 2.5 | Graduate school | 5.4 | 3.3 | ||
| 18-35 | 26.1 | 16.1 | Unknown | 24.2 | 31.7 | ||
| 36-49 | 36.1 | 34.2 | Employment (%) | ||||
| 50-65 | 28.4 | 36.6 | Employed | 54.8 | 44.6 | ||
| 65+ | 5.3 | 9.4 | Not employed | 28.2 | 36.4 | ||
| Male (%) | 58.8 | 60.6 | Retired | 6.2 | 9.1 | ||
| Race, Ethnicity (%) | Unknown | 10.8 | 9.9 | ||||
| White | 76.8 | 64.4 | Insurance (%) | ||||
| African American | 17.3 | 28.9 | Medicare only | 16.1 | 25.7 | ||
| Asian | 4.1 | 4.8 | Medicaid only | 5.6 | 5.7 | ||
| Other | 1.8 | 1.9 | Private | 55.0 | 33.1 | ||
| Hispanic/Latino | 13.2 | 12.2 | Medicare + Private | 9.9 | 14.1 | ||
| Blood Type (%) | Other or missing | 13.4 | 21.4 | ||||
| A | 35.8 | 38.0 | Cause of ESRD (%) | ||||
| B | 14.1 | 12.4 | GN | 29.9 | 24.0 | ||
| AB | 3.1 | 5.2 | Diabetes | 20.9 | 22.3 | ||
| O | 47.0 | 44.2 | Hypertension | 16.0 | 21.3 | ||
| Average income | 32,482 | 30,137 | Other | 33.2 | 32.4 | ||
| Time on waiting list (days) | 292 | 563 | OPO median waiting time (days) | 397 | 357 | ||
Table VI-4. Distribution of Living vs. Deceased Donor Kidney Transplants Among Recipient Characteristics, 1995-2000 (Row Percent)
| Measure | Living | Deceased | Measure | Living | Deceased |
|---|---|---|---|---|---|
| Transplants | Education (%) | ||||
| Sample size (n) | 10,331 | 38,797 | None | 14.0 | 86.0 |
| Percent | 21.0 | 79.0 | 0-8 years | 17.4 | 82.6 |
| Age (years, %) | High school grad | 20.3 | 79.7 | ||
| 0-9 | 20.0 | 80.0 | College | 26.5 | 73.5 |
| 10-17 | 23.9 | 76.1 | Graduate school | 30.2 | 69.8 |
| 18-35 | 30.1 | 69.9 | Unknown | 21.7 | 78.3 |
| 36-49 | 21.9 | 78.1 | Employment (%) | ||
| 50-65 | 17.1 | 82.9 | Employed | 24.7 | 75.3 |
| 65+ | 13.1 | 86.9 | Not employed | 17.1 | 82.9 |
| Male (%) | <20.5 | 79.5 | Retired | 15.4 | 84.6 |
| Female (%) | 21.8 | 78.2 | Unknown | 22.5 | 77.5 |
| Race, Ethnicity (%) | Insurance (%) | ||||
| White | 24.1 | 75.9 | Medicare only | 14.3 | 85.7 |
| African American | 13.8 | 86.2 | Medicaid only | 20.7 | 79.3 |
| Asian | 18.4 | 81.6 | Private | 30.6 | 69.4 |
| Other | 19.9 | 80.1 | Medicare + Private | 15.8 | 84.2 |
| Hispanic/Latino | 22.8 | 77.2 | Other | 21.1 | 78.9 |
| Blood Type (%) | Cause of ESRD (%) | ||||
| A | 20.1 | 79.9 | GN | 25.2 | 74.8 |
| B | 23.3 | 76.8 | Diabetes | 20.2 | 79.8 |
| AB | 13.6 | 86.4 | Hypertension | 16.8 | 83.2 |
| O | 22.0 | 78.0 | Other | 21.6 | 78.4 |
The influence of race on patient survival is complex. Asians exhibit the lowest death rates on the waiting list[Table 5.3] and slightly higher survival rates following both Deceased Donor [Table 5.9a] and Living Donor[Table 5.9b] transplantation. Whites, in contrast, have the highest death rates while on the waiting list. White and African American recipients have approximately equivalent five-year survival rates following transplantation from deceased or living donors (Table VI-5)Deceased Donor [Table 5.9a]/Living Donor[Table 5.9b]. This observation, while consistent over the last decade, remains unexplained.
Overall patient survival at five years is 90% following living donor transplantation, compared to 80% following transplantation from a deceased donor[Table 5.9]. The degree of HLA match does not appear to have an important effect on patient survival for either type of transplant. Recipients of deceased donor kidneys from donors aged 65 and over have poorer survival (65% compared to 80% overall)[Table 5.9a]. Death rates posttransplant are consistently highest among recipients with diabetic kidney disease[Table 5.7], and diabetic recipients have the poorest five-year patient survival following transplantation from deceased donors (68% versus 80% overall) or living donors (81% versus 90% overall)[Table 5.9].
Table VI-5. Five-Year Patient Survival (%), by Race and Donor Type
| Race | Donor Type | |
|---|---|---|
| Deceased | Living | |
| White | 79.7% | 89.8% |
| Asian | 87.1% | 95.0% |
| African American | 79.4% | 88.1% |
The 2002 OPTN/SRTR Annual Report includes data on three different cohorts of recipients. Three-month and one-year graft survival is reported for recipients transplanted in 1999-2000, three-year graft survival is for those transplanted from 1997-1998, and five-year outcomes are for those transplanted between 1995-1996. The advantage of this “cohort shift” technique is that it reports actual, rather than estimated, graft survival. Given the relationship between short- and long-term outcomes, the finding that one-year graft survival for living or deceased donor kidney recipients has improved by only 1%-2% since 1995 imparts validity to this statistical approach [Table 1.12].
Overall, one- and five-year allograft survival rates for recipients of deceased donor kidneys were 88% and 63%, respectively[Table 5.8a]. Gender, ABO blood group, and transplant center volume exerted little impact on short- or long-term graft survival. Although retransplantation confers a consistent decrement (2%-4%) in outcomes at all time points, the magnitude of the difference has grown smaller in more recent years (20), likely a result of improved crossmatching and more effective immunosuppression.
One-year graft survival rates among adult recipients were best for those aged 35-49 (90%) and worst for those aged 65 or older (84%). Similarly, five-year graft survival rates were 66% for recipients aged 35-49 and 53% for recipients 65 and older. Recipients between the ages of 18 and 49 demonstrated one-year and five-year graft survival of roughly 90% and 64%, respectively. Poorer short- and long-term outcomes are demonstrated in recipients above and below these age groups. For recipients older than 50, the decrement in graft survival is accompanied by a decrement in patient survival, implying significant losses due to death with function[Table 5.8a]. Alternatively, younger recipients appear to lose kidney function over time without losing their lives, implying a greater impact from allograft losses due to other causes (Figure VI-14).
Racial and ethnic backgrounds appear to exert little impact on graft survival during the first year following transplantation. Beyond one year, however, African American patients fare worse than patients of all other ethnic backgrounds. By five years after transplantation, 72% of Asian recipients maintain functioning grafts, compared to only 54% of African American recipients. Recipients of white race and Hispanic/Latino ethnicity demonstrate intermediate survival of approximately 66%. These findings have been quite consistent over the last decade, and, though not easily explained, likely result from an adverse mix of socioeconomic and immunologic risk factors for graft loss (21).
In keeping with previous reports, one- and five-year deceased donor graft survival is best in recipients with polycystic kidney disease (92% and 74% respectively). Decrements in five-year graft survival (4%-7%) are noted for patients with diabetes, hypertensive nephrosclerosis, and other vascular diseases. As a group, ESRD patients with polycystic kidneys fare better regardless of therapeutic modality, a finding thought to represent the lack of comorbid complications in many of these patients (5).Donor age exerts significant effects on outcomes after transplantation: Kidneys from very young and very old donors fare worse. While some differences are apparent at three months and one year following transplantation, the impact is greatest on long-term outcomes (Figure VI-15). Delayed graft function, defined as dialysis within the first week after transplantation, continues to exert a strong negative effect on one-year graft survival (82% for those who needed dialysis within the first week, 93% for those who did not) and five-year allograft survival (51% and 69%, respectively)[Table 5.8a]. These findings, with greater impact on long-term than short-term graft survival, are consistent with recent theories linking chronic allograft failure to limited repair capacities of injured tissues (22).
Immunologic variables remain important. Somewhat surprisingly (given recent advances in crossmatch techniques) there remains a strong influence of presensitization on graft survival at one and three years following transplantation, and five-year survival is 64% for unsensitized recipients versus 53% for those with PRA >80%[Table 5.8a](23). HLA matching continues to influence graft outcomes, with greater differences between well-matched and poorly-matched recipients as time after transplantation lengthens. As can be seen in Figure VI-16, only completely matched and completely mismatched recipients differ substantially from the mean, supporting current mandatory sharing of zero-mismatched kidneys (13). These data also offer support for proposals to discourage six-antigen mismatches in allocation policies (24).
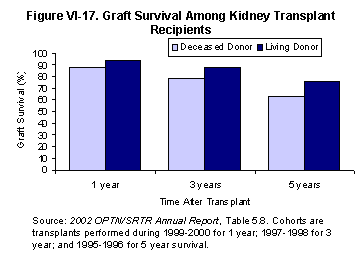
Recipients of living donor kidneys continue to fare better than recipients of kidneys from deceased donors (Figure VI-17).Overall, most recent one-year allograft survival was 94% and five-year survival was 76%. One-year allograft survival ranged from 100% for recipients less than 1 year old to 92% for recipients 65 and older. Similarly, five-year allograft survival ranged from 92% in recipients less than one year old to 66% for recipients 65 and older [Table 5.8b].
The effects of various demographic and transplant-related variables on living donor kidney outcomes are similar to those among transplants from deceased donors. Little difference was observed regarding gender and blood type. The well-described adverse effect of prior transplantation also occurred in the living donor population, increasing over time to a difference in graft survival of 8% at five years. Asian recipients had the best graft survival rate at both one and five years, 96% and 84%, respectively; while African Americans had the worst graft survival at these time points (93% and 64%, respectively). Among etiologies of ESRD, the best five-year kidney survival was found in patients with polycystic kidney disease (84%). The worst outcomes were associated with patients with hypertensive nephrosclerosis (70%), retransplant/graft failure (67%), and renovascular and other vascular disease (67%)[Table 5.8b].
Dialysis within the first week after transplantation bodes worse for living donor kidney recipients than for cadaveric kidney recipients, with a differential effect of 23% after five years[Table 5.8b]. The association between donor age and graft survival is weaker among living donor recipients than deceased donor recipients. Kidneys from living donors age 65 and over have moderately lower survival (82%) after three years, but this compares favorably with the 89% survival rate for kidneys from much younger (age 18-34) deceased donors. These statistics likely reflect the same variable: prospective evaluation of renal function in living donors, an opportunity not afforded in the deceased donor process (25). Thus, whereas increasing age in a deceased donor kidney may correlate with reduced organ quality, older living donors who successfully complete the evaluation process are much more likely to demonstrate normal or near-normal renal function. Accordingly, delayed graft function in a living donor kidney is much more likely to represent immunologic insult rather than underlying organ quality or ischemic injury, with a more pronounced impact on graft survival than might occur with a kidney from a deceased donor.
Living donor source does not appear to influence allograft outcomes until five years, at which point transplants among siblings demonstrate the best survival (81%) [Table 5.8b]. This likely represents the favorable influence of the two-haplotype match on sibling transplant outcomes. Unrelated living donor transplants have the lowest five-year graft survival, at 72%. Progressive differences in transplant survival resulting from HLA mismatch become evident at one year, with a 5% spread between zero-mismatch and six-mismatch transplants. By five years this difference reached 18%, with the largest decrement of 9% survival difference noted between zero-mismatch and one-mismatch transplants. Nonetheless, these data point to the benefits of living donor transplantation, with even the poorest outcomes at five years comparable to or exceeding the best outcomes possible with transplants from deceased donors.
The number of patients awaiting SPK transplants rose from 751 in 1992 to 2,503 in 2001[Table 8.1]. Over the same period, the average age of those on the waiting list rose from 36.9 to 41.1 years, with 16% at least 50 years old by 2001. Whites made up the majority of registrants throughout the decade (81% in 2001), a finding discussed by Isaacs et al. (26). Over the same period, the percentage of African Americans on the waiting list rose from 10% to 16%, and the percentage of Hispanic/Latino registrants rose from 1% to 8%. There was little change in the gender distribution of the waiting list over the last 10 years, with women making up 42%-45% of wait-listed patients. Between 1992 and 2001, the percentage of patients on the kidney-pancreas waiting list awaiting SPK retransplantation remained constant at 2-4%.
As is the case with other organs, growth in the numbers of patients awaiting SPK transplantation has far outpaced the growth in the number of transplants being performed, resulting in a dramatic increase in time spent on the waiting list. The percentage of registrants on the waiting list longer than a year rose from 24% in 1992 to 50% in 2001 (including 20% waiting longer than two years). These times remain relatively short, however, compared to those for patients awaiting kidney transplantation [Tables 5.1 and 8.1].
For candidates wait-listed in 1994, median time to SPK transplantation was 255 days. Since then, it has more than doubled to 546 days for those listed in 2000[Table 8.2]. Minority registrants (particularly Hispanics/Latinos and African Americans) wait longer for SPK transplantation than whites, but the difference in waiting time is much less pronounced than in kidney transplantation alone[Tables 5.2 and 8.2]. Likewise, those with ABO blood types O and B wait longer for transplantation.
Given the important survival advantages of early transplantation for diabetics with ESRD, these trends are disturbing (27). Prolonged waits for transplantation not only compromise subsequent graft and patient survival, but have other consequences as well. Unlike death rates on the kidney waiting list, which have remained relatively stable over the past decade, death rates among those awaiting SPK transplantation have increased by about 18% since 1993[Table 8.3].
Despite the ominous trends in waiting list demographics, outcomes for recipients of SPK transplants continued to improve dramatically[Table 8.7]. The annual death rate (per 1,000 patient years at risk) for recipients 35-49 years old declined from 103 in 1993 to 42 in 2001. Similar drops were evident for recipients in other age groups. Variables that appeared to increase death rates are increasing age, a history of prior organ transplantation, and elevated PRA.
Patient survival at one year after SPK transplantation increased from 93% in 1991 to 95% for those transplanted in 2000[Table 1.13]. Five-year patient survival is now 83%[Table 8.9]. No demographic variables exert a noticeable effect on patient survival up to three years beyond SPK transplantation. However, by five years, recipients older than 50 demonstrate substantial reduction in patient survival (75%, compared to 82% for recipients aged 35-49 at transplantation).
Overall, one-year and five-year kidney graft survival rates for SPK transplant recipients were 92% and 73%, respectively[Table 8.8]. This compares favorably with graft survival rates of 88% and 63% for recipients of deceased donor kidney transplants alone[Table 5.8a]. Gender, age, blood type, and having received a previous transplant did not appear to affect either short- or long-term kidney graft survival. African Americans had worse long-term kidney graft survival (64% five-year kidney graft survival) than other racial groups (74% for whites; 75% for Asians), a trend similar to that seen among recipients of kidney transplants alone. Not surprisingly, organs from donors older than 35 were associated with reduced five-year kidney graft survival (donors aged 35-49: 65%; donors aged 50-64 years: 70%), in contrast to donors younger than 35. Center volume and level of HLA mismatch did not influence short- or long-term kidney graft survival.
Overall, one-year and five-year pancreas graft survival rates for kidney-pancreas transplant recipients were 84% and 69%, respectively[Table 8.8]. Interestingly, long-term pancreas graft survival rates between whites (70%) and African Americans (65%) were relatively similar. Neither gender, nor blood type, exerted any marked effect on pancreas graft survival. Older donor age was associated with a worse five-year pancreas graft survival, though not to the same extent as with kidney grafts. Pancreas graft survival appears to be independent of HLA matching, although the data are variable and difficult to interpret (29).
In 2001, there were 1,089 patients on the waiting list for an isolated pancreas transplant — 63% for PAK, 37% for PTA[Tables 6.1 and 7.1]. Ten years ago there were only 139 candidates on this waiting list. Most of this increase came from the growing number of PAK candidates (up twelvefold from 1992 to 2001). The number of PTA candidates increased fivefold over the same period. SPK candidates still comprise the largest proportion of the waiting list, with 2,503 awaiting transplantation in 2001 [Table 8.1]. However, this represents only a threefold increase from 10 years ago, indicating increasing acceptance of PAK and PTA procedures, as well as dramatic growth in the number of centers offering isolated pancreas transplantation (from 20 in 1992 to 79 in 2001) (26,27).
Of the 1,089 candidates waiting for an isolated pancreas transplant in 2001, 21% were 18-34 years old, 63% were 35-49, and 15% were 50-64. This represents a shift in the last 10 years toward older candidates; a trend that was present in both the PAK and PTA categories. In 1992, the percentages for these age groups were 42%, 52%, and 5%, respectively. Most of those currently awaiting transplantation are white, with fewer than 10% of PTA and PAK candidates from minority populations. These proportions have not changed significantly since 1992. In 2001, women made up 54% of the PTA waiting list and 44% of the PAK waiting list.
Overall, the number of candidates on the waiting lists who have undergone a previous pancreas transplant did not increase significantly since 1992. Of the 403 PTA candidates in 2001, only 7% had undergone a previous pancreas transplant. Alternatively, 24% of the 686 PAK candidates had undergone a previous SPK transplant and experienced subsequent pancreas graft failure, a decrease from 27% in 1992. This decline in percentage awaiting retransplantation reflects both better outcomes for SPK transplants and increasing demand for solitary pancreas transplantation.
Average time on the waiting list differs between PAK and PTA candidates. At the end of 2001, 68% of PAK candidates had been on the list for three months to two years, and 13% had been waiting longer than two years. In 1996, 24% had been waiting for longer than two years. Several factors probably contributed to this change, including rapid growth in those desiring PAK transplantation, more centers offering the procedure, and a new trend to proceed with pancreas transplantation sooner after kidney transplantation than was previously thought optimal (28). With PTA candidates, however, 32% of PTA candidates in 2001 had been on the waiting list for longer than two years, an increase from 20% in 1992 — again reflecting slower growth in this field.
Death rates for those awaiting PTA and PAK transplants have fluctuated over the last 10 years but rose steadily over the last four years for PAK candidates, perhaps reflecting changes in criteria for defining candidates, with more older patients on the transplant list[Tables 6.3 and 7.3].
The median time to transplant in 2000 was shorter for PTA registrants (343 days) than for PAK registrants (437 days) or SPK registrants (546 days) [Tables 6.2, 7.2, and 8.2]. For SPK registrants, time to transplant has steadily increased since 1994. However, for both PTA and PAK registrants, time to transplant steadily decreased between 1995 and 1999. In 2000, however, median time to transplant for these two categories rose, perhaps reflecting increasing demand. Time to transplant did not differ by age or gender, and remained longest for registrants with blood type O. In 2000, for a PTA transplant, the median time to transplant was 409 days for registrants with blood group O, compared to 189 days for blood group AB [Table 6.2].
The most noticeable trend among pancreas recipients is the rapid growth in numbers of procedures performed, especially since Medicare deemed pancreas transplants reimbursable in 1999. A total of 163 PTAs were performed in 2001, up substantially from 34 in 1992 and 134 in 1999. The number of PAK transplants increased to an even greater extent: 305 were performed in 2001, up from 27 in 1992 and 221 in 1999. In contrast, the number of SPK transplants in 2001 was 885, less than twice the number performed in 1992 and down 5% from the 933 transplants in 1999. Although SPK transplants still accounted for 65% of pancreas transplants performed in 2001, this percentage is a marked change from 89% in 1992 (Figure VI-18) [Tables 6.4 and 7.4].
Although most pancreas transplants in 2001 were performed in recipients aged 18-49, an increasing percentage of the recipients were older. For PTA recipients, the 50-64 age group accounted for 16% of PTA recipients, up from only 3% in 1992. Similarly, the 50-64 age group accounted for 12% of the PAK recipients in 2001, up from 7% in 1993. Of all isolated pancreas transplants in 2001, whites received 91% and African Americans received 7%. Of the 305 PAK recipients, 25% had previously undergone an SPK transplant.
For PTA recipients, patient survival was 99% at one year (1999-2000 cohort), 86% at three years (1997-1998 cohort), and 78% at five years (1995-1996 cohort). Other variables, such as sex, donor age, level of HLA mismatching, and transplant center volume, did not seem to affect patient survival [Tables 6.8, 6.9, 7.8, and 8.9].
For these same cohorts, graft survival was 81% at one year, 57% at three years, and 32% at five years (Figure VI-19). For these patients, recipient age or gender did not affect graft survival trends. Donor age is an important variable influencing outcomes. One-year graft survival was 84% for transplants from donors aged 18-34, 77% from donors aged 35-49, and 54% from donors aged 50-64. HLA matching also seemed to have some impact, with declining graft survival rates according to match grade becoming evident even after one year (92% for one mismatch, 66% for five mismatches).
For PAK recipients [Tables 7.8 and 7.9], patient survival was 96% at one year, 89% at three years, and 77% at five years, and, again, was equivalent in males and females. Other variables such as donor age, level of HLA matching, and transplant center volume did not seem to affect patient survival rates. For PAK transplants, recipient age or gender also did not seem to affect graft survival. In contrast to the PTA category, donor age and HLA matching did not seem to affect graft survival rates.
Pancreas graft survival in the short term (three months) was fairly similar for all three transplant categories (Figure VI-19). However, by five years after transplantation, the groups exhibited significant differences in graft survival. The best long-term graft survival rates were seen in SPK recipients, and worst were seen in PTA recipients. The divergence is likely a reflection of the increased immunologic problems (acute and chronic rejection) still seen with isolated pancreas transplants (26).
Kidney transplantation accounted for 59% of all organ transplants in 2001. Evaluation of trends revealed the following key changes over the last decade:
The number of simultaneous kidney-pancreas (SPK) transplants has remained static since 1995, but the waiting list has doubled in size. Rapid growth was observed in the numbers and success rates of isolated pancreas transplantation.
The following individuals prepared this chapter: Robert S. Gaston, MD1, Denise Y. Alveranga, MD2, Bryan N. Becker, MD3, Dale A. Distant, MD4, Philip J. Held, PhD5, Jennifer L. Bragg- Gresham, MS5, Abhinav Humar, MD6, Alan Ting, PhD7, James J. Wynn, MD8, Alan B. Leichtman, MD9. 1University of Alabama at Birmingham; 2Lifelink Transplant Institute Tampa; 3University of Wisconsin; 4State University of New York Health Science Center at Brooklyn; 5SRTR/URREA; 6University of Minnesota Twin Cities; 7OPTN/UNOS; 8Medical College of Georgia; 9SRTR/University of Michigan.
1. Wolfe RA, Ashby VB, Milford EL, et al. Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med 1999;341:1725-1730.
2. Gaston RS. Evolution of Medicare policy involving transplantation and immunosuppressive medications: past developments and future directions. In: Field MJ, Lawrence RL, Zwanziger L, editors. Extending Medicare Coverage for Preventive and Other Services. Washington, DC: National Academy Press; 2000. p. D23-D38.
3. Kasiske BL, Cangro CB, Hariharan S, et al. The evaluation of renal transplant candidates: clinical practice guidelines. Am J Transplantation 2001;2 (Suppl. 1):5-95.
4. Kasiske B, Ramos E, Gaston R, et al. The evaluation of renal transplant candidates: clinical practice guidelines. J Am Soc Nephrol 1995;6:1-34.
5. United States Renal Data System (USRDS). 2001 Annual Data Report. Minneapolis, MN; 2001.
6. Schweitzer EJ, Wilson J, Jacobs S, et al. Increased rates of donation with laparoscopic donor nephrectomy. Ann Surg 2000;232:392-400.
7. Matas AJ, Garvey CA, Jacobs CL, Kahn JP. Nondirected donation of kidneys from living donors. N Engl J Med 2000;343:433-436.
8. Deierhoi MH, Barger BO, Hudson SL, Shroyer TW, Diethelm AG. The effect of erythropoietin and blood transfusions on highly sensitized patients on a single cadaver renal allograft waiting list. Transplantation 1992;53:363-368.
9. Danovitch GM, Hariharan S, Pirsch JD, et al. Management of the waiting list for cadaveric kidney transplants: Report of a survey and recommendations by the Clinical Practice Guidelines Committee of the American Society of Transplantation. J Am Soc Nephrol 2002;13:528-535.
10. Wolfe RA, Ojo AO, Ashby VB. Analyses for data request from the Minority Affairs Committee of 4/5/2001. Ann Arbor, MI: Scientific Registry of Transplant Recipients (SRTR); 2001 10/12/01.
11. Gaston RS. Addressing minority issues in renal transplantation: is more equitable access an achievable goal? Am J Transplant 2002;2:1-3.
12. Nelson PW, Shield CF, Muruve NA, et al. Increased access to transplantation for blood group B cadaveric waiting list candidates by using A2 kidneys: time for a new national system? Am J Transplant 2002;2:94-99.
13. Takemoto SK, Terasaki PI, Gjertson DW, Cecka JM. Twelve years' experience with national sharing of HLA-matched cadaveric kidneys for transplantation. N Engl J Med 2000;343:1078-1084. 14. Schweitzer EJ, Yoon S, Hart J, et al. Increased living donor volunteer rates with a formal recipient family education program. Am J Kidney Diseases 1997;29:739-745.
14. Schweitzer EJ, Yoon S, Hart J, et al. Increased living donor volunteer rates with a formal recipient family education program. Am J Kidney Diseases 1997;29:739-745.
15. Terasaki PI, Cecka JM, Gjertson DW, Takemoto S. High survival rates of kidney transplants from spousal and living unrelated donors. N Engl J Med 1995;333:333-336.
16. Meier-Kriesche H-U, Port FK, Ojo AO, et al. Effect of waiting time on renal transplant outcome. Kidney Int 58: 1311-1317, 2000
17. Schaubel D, Desmeules M, Mao Y, Jeffery J, Fenton S. Survival experience among elderly end-stage renal disease patients. A controlled comparison of transplantation and dialysis. Transplantation 1995;60:1389-1394.
18. Ojo AO, Hanson JA, Meier-Kriesche HU, et al. Survival in recipients of marginal cadaveric donor kidneys compared with other recipients and wait-listed transplant candidates. J Am Soc Nephrol 2001;12:589-597.
19. Zenios SA, Wein LM, Chertow GM. Evidence-based organ allocation. Am J Medicine 1999;107:52-61.
20. Cecka JM. The UNOS Scientific Renal Transplant Registry. In: Cecka JM, Terasaki PI, editors. Clinical Transplants 1998. Los Angeles: UCLA Tissue Typing Laboratory; 1999. p. 1-16.
21. Young CJ, Gaston RS. Renal transplantation in black Americans. N Engl J Med 2000;343:1545-1552.
22. Halloran PF, Melk A, Barth C. Rethinking chronic allograft nephropathy: the concept of accelerated senescence. J Am Soc Nephrol 1999;10:167-181.
23. Karpinski M, Rush D, Jeffery J, et al. Flow cytometric crossmatching in primary renal transplant recipients with a negative anti-human globulin enhanced cytotoxicity crossmatch. J Am Soc Nephrol 2001;12:2807-2814.
24. Schnitzler MA, Hollenbeak CS, Cohen DS, et al. The economic implications of HLA matching in cadaveric renal transplantation. N Engl J Med 1999;341:1440-1446.
25. Kasiske BL, Ravenscraft M, Ramos E, Gaston RS, Bia MJ, Danovitch GM. The evaluation of living renal transplant donors: clinical practice guidelines. J Am Soc Nephrol 1996;7:2288-2313.
26. Humar A, Ramcharan T, Kandaswamy R, et al. Pancreas after kidney transplant. Am J Surg 2001;182:155-161.
27. Hariharan S, Pirsch JD, Lu CY, et al. Pancreas after kidney transplantation. J Am Soc Nephrol 2002;13:1109-1118.
28. Humar A, Sutherland DER, Ramcharan T, Gruessner RW, Gruessner AC, Kandaswamy R. Optimal timing for a pancreas transplant after kidney transplant. Transplantation 2000;70:1247-1250.
29. United Network for Organ Sharing. OPTN/UNOS policy 3.5. Allocation of cadaveric kidneys. Richmond, VA; 2002.
Center-Specific Reports | OPO Reports | National Reports | Data Accuracy | Report Timeline
Home | Who We Are | What We Do | About Transplants | Transplant Statistics | Research Resources | Contact Us | Site Map
SRTR Site designed and maintained by Diamond Bullet Design & URREA.