
|

|
|
Transplant Statistics: Annual Report : Appendix H
Both the OPTN and the SRTR serve the transplant community, government officials, researchers, and the public by providing data and conducting studies that further the advances in organ procurement and transplantation. Within the guidelines of each contractor's purview, this is accomplished by providing specific data to support the policy development activities of the OPTN, the Secretary's Advisory Committee on Organ Transplantation (ACOT), and other governmental agencies as requested; making summary statistics or certain data files available for researchers, patients, and others in the public and private sector; and conducting original research in response to questions from transplant professionals. Recent analytic and dissemination activities are outlined below.
The primary function of the standing and ad hoc OPTN/UNOS committees is to provide initial review and analysis of policy proposals, revisions, and initiatives before presentation to the OPTN/UNOS Board of Directors. As the OPTN and SRTR contractors, UNOS and URREA staff assist the committees with the analytical research and support necessary for policy consideration and development. This support entails providing technical, operational, and statistical analyses on the wide variety of organ procurement and transplantation issues facing the OPTN/UNOS committees.
This section describes the ways the OPTN and SRTR each has provided these types of analytical support over the past year.
The UNOS Information Technology (IT) Department develops and supports all computer systems involved in OPTN organ allocation and data collection efforts. When changes to policies necessitate changes to these systems, Detailed Specification Documents are written to identify the specific system revisions to be programmed in accordance with the features of each Board-approved policy change, Alternate Point Assignment allocation system, Alternative Local Unit, and sharing agreement.
As these computer systems have grown more complex in recent years, and increasingly intertwined with policy development, IT has assumed a greater role in the support of the committees charged with policy development. This broadened involvement of IT from policy implementation to now policy development began with the intensive year-longeffort leading to the approval and implementation of the MELD/PELD system for liver allocation. UNOS IT staff now attend each meeting of the organ allocation committees and work closely with the committee, or its technical subcommittee, to ensure that what eventually becomes a proposal for the Board's consideration may be fully implementable if approved. As a new policy takes shape, IT provides a Detailed Specification Document that reflects how the policy as proposed would be implemented. The process then may take on an iterative nature as the technical analyses may lead the committee or subcommittee to further refine its proposal.
Upon receiving OPTN data, IT specialists at the SRTR process and enrich the data, which allows the SRTR to perform statistical analyses in support of the OPTN/UNOS committees. This data processing includes checking the data for consistency and accuracy, as well as linking the data to other sources for extra ascertainment of death. These important functions, performed by the SRTR IT specialists, are valuable in supporting the research and policy development of OPTN/UNOS committees and serve as additional validation of OPTN data.
The Policy Compliance Department (PCD) actively monitors the extent to which members are able to adhere to organ allocation and data submission policies. PCD
provides compliance analyses on both a routine and ad hoc basis and conducts various projects in support the committees. These projects may be summarized generally in two areas: 1) referral of policy clarifications to the applicable OPTN/UNOS committee, and 2) development and testing of online policy implementations.
Policy Clarification. PCD on-site reviewers encounter many policy issues and questions from members and on-site reviews. These may include varying policy interpretations as well as areas not addressed by current policy. These issues are summarized and forwarded to the applicable OPTN/UNOS committees for resolution. Since January 2002, the PCD has sent one issue to the Liver/Intestine Transplantation Committee, three issues to the Kidney/Pancreas Transplantation Committee, and one issue to the Organ Procurement Organization (OPO) Committee.
One example of the interaction between PCD and the committee system can be seen in the revision of policy for Justification Form Submission. After several on-site reviews, PCD staff acknowledged that most members were not in compliance with Justification Form Submission within 24 hours of listing. PCD staff forwarded this information to the UNOS Liver and Thoracic Committees for review. Based on those data, the Liver and Thoracic Committees recommended that policies regarding justification form submission be changed from 24 hours to one business day. The UNOS Board of Directors later approved this.
Online Development and Testing of Policy Implementations. PCD staff coordinates with IT staff in the development, testing, and electronic implementation of policy changes to insure that new applications adhere to current policies. For example, PCD staff played an intricate role in the development of the online justification form submission, as well as the online peer review process of urgent status heart and liver listings. (See Appendix F for more information about PCD activities.)
Almost all of the OPTN/UNOS committees rely heavily on the statistical analysis of trends in transplantation to inform their policy recommendations. Per contractual specifications, the OPTN contractor provides descriptive data and the SRTR contractor provides inferential statistics in support of policy development. Recent statistical support activities by each contractor are detailed separately for each of the committees listed below.
OPTN. OPTN staff provided an analysis of delinquent data collection forms by organ type and transplant program. This analysis included an assessment of the anticipated workload reduction for each program if certain expected follow-up forms were not required. Based on these data and analyses, the DAC proffered an "amnesty policy" that temporarily eases the requirements for submission of posttransplant follow-up forms.
To assess the current completeness of various follow-up data, the DAC requested an extensive investigation of the rate at which patients are lost to follow-up after receiving a transplant. This analysis was provided for kidney, liver, kidney-pancreas, heart, and lung programs both cumulatively to date and by year. Specific topics addressed included organ-specific differences in lost to follow-up rates, differences in rates according to graft function, and differences in rates according to program volume. An additional analysis in this area ascertained the impact on overall and program-specific survival rates when the National Death Index (NDI) database was used to determine the status of patients lost to follow-up to the OPTN.
Overall lost to follow-up rates for the period from October 1, 1987, to December 31, 1999, ranged from 2.7% of lung transplants to 16.7% of kidney transplants. Among kidney and kidney-pancreas transplants, the lost to follow-up rate was greater among patients with failed grafts. There was very little correlation between transplant program volume and lost to follow-up rates for all organs. When follow-up data were supplemented with data from the NDI, overall survival rates were very similar to those produced in absence of the supplemental information, indicating that, in general, the mortality risk of patients lost to follow-up is similar to that of patients who are not lost to follow-up by the OPTN.
OPTN. In order to determine if the degree of sensitization in patients listed for a retransplant correlated with the level of HLA mismatch of the first transplant, the Histocompatibility Committee requested data showing the number and frequency of sensitized and unsensitized patients relisted for a second kidney transplant from 1995 to 2001, stratified by the degree of HLA mismatch of their first transplant. Additionally, changes in PRA between initial listing and relisting were requested. There appeared to be a correlation between the number of A,B mismatches and percentage of sensitized patients. A second set of requested statistical analyses sought to determine if in OPOs with variances that do not use HLA points, minorities are transplanted at equal, or near equal, rates as whites.
A third set of analyses conducted for the Committee addressed the ongoing monitoring of the time-limited voluntary allocation variance involving the use of cross-reactive antigen groups (CREGs) to prioritize renal waiting list candidates for available donors. The data showed that in the CREG arm, the CREG matched grafts had a significantly higher two-year survival (93%) than the CREG mismatched grafts (80%). There was no significant difference in the percentage of minorities transplanted between the CREG and control arms. A fourth set of analyses was performed to determine the discrepancy rate for HLA typing of deceased donors, comparing the typings entered for the match run with typings entered in the donor and recipient histocompatibility forms. The data showed that the HLA typing discrepancy rate for typings performed between 1995 and 2001 was 5%, considerably lower than the 14% discrepancy rate seen in the 1987-1995 data. Finally, at the Committee's request OPTN staff provided descriptive statistics to determine if there is over-reporting of B15 and B40 for deceased donors (1998-2002) by molecular methods, which may represent incorrect serologic assignments for some B15 and B40 alleles.
SRTR. The Histocompatibility Committee requested analyses to investigate graft loss and rejection episodesamong highly sensitized patients, by technique used for the final crossmatch. The SRTR looked specifically at three populations: 1) outcomes where there were no positive crossmatches, 2) outcomes by looking at specific flow and AHG positive and negative combinations, and 3) outcomes by looking at specific flow and AHG, B and T, positive and negative combinations. By final crossmatch technique and PRA category there was a tendency towards fewer first rejections when the final crossmatch technique was performed by flow cytometry than when it is performed by AHG. Similarly, there was a trend in rejection episodes and graft survival favoring final crossmatch technique of flow cytometry. A result of B positive / T negative by flow cytometry was not a better predictor of percentage or rate of a first rejection episode than was B negative / T negative, but there was a trend in this direction with higher PRA and by adjusted and unadjusted odds ratio. Both may be better than AHG alone, presumably reflecting exclusion of AHG negative, flow cytometry T positive candidates. There was a trend towards a reduced risk of graft failure when B negative / T negative was contrasted with B positive / T negative. Graft survival differences between these categories may increase over time.
To investigate both long- and short-term graft survival, the Committee was also interested in 3-, 5-, and 10-year kidney graft survival by specific groupings of HLA antigens and by CREGs. For all analyses, the data were stratified by ethnicity and first graft or regraft. Survival differences were observed among the different HLA groups, as well as the CREG groups. Survival differences were seen among the race/ethnicity groupings. The degree of matching and graft survival also appeared to differ among the race/ethnic groups. This analysis supports the finding that the DR loci are very important to graft survival.
OPTN. The IR Committee has continued to monitor transplant program compliance with the OPTN guidelines regarding the transplantation of deceased donor organs into nonresident aliens. The OPTN has provided the Committee with the data needed to identify, and review trends, at those centers where deceased donor transplants to nonresident aliens make up more than 5% of the total deceased donor transplants performed during a year. Data also were provided to investigate instances when programs may be in geographic areas that recover a large number of nonresident alien deceased donors.
OPTN. A primary charge of the DWG is to recommend to the DAC changes for the OPTN data collection forms to increase their utility and efficiency. To facilitate this process, OPTN staff provided a detailed list of every element collected on the current forms, classified according to their use in OPTN functions, contractual requirements, Final Rule requirements, research, or internal functions. To assess the capacity of the current data elements to measure resource utilization, the DWG requested a preliminary validation analysis on the accuracy of posttransplant length of stay in the OPTN database with the center's records. Finally, the DWG requested information on fields that have a 10% or greater missing or "unknown" response as part of a broad assessment of data timeliness and completeness.
SRTR. The DWG requested that the SRTR address the issue of health resource utilization as measured by hospital length of stay (LOS). The major variables of interest were pretransplant, posttransplant, and total days in hospital for the first transplant occurring during the hospitalization period. For the initial inquiry, the SRTR looked at hospital LOS for all patients, patients who died in the hospital, and patients alive at discharge. The analysis covered recipients of heart-lung, heart, intestine, kidney, kidney-pancreas, liver, lung, and pancreas transplants. In addition, the SRTR examined the mean pretransplant days for transferred heart and liver patients, as well as the LOS for heart and liver patients by medical urgency status. The second LOS analysis performed by the SRTR examined the average facility median LOS for all centers, transplant centers below the median number of transplants for all centers, and centers at or above the median number of transplants for all centers. In addition, the percentage of patients retransplanted, transferred, or with Medicare coverage as their primary or secondary source of payment were reported.
In an effort to determine the validity and predictive value of donor kidney biopsy data, the Data Working Group requested and reviewed an SRTR analysis of these data. While only a small fraction of donor kidneys are actually biopsied, for those that are, any degree of glomerulosclerosis was found to be a significant predictor of graft survival. These data were deemed worthwhile to continue collecting because of their potential value as additional expanded donor criteria.
Several data items flagged for removal by the various organ subcommittees of the DWG were evaluated for possible deletion or revision. The SRTR provided feedback to the DWG committee on several specific data items from the Transplant Candidate, Registration, and Cadaver Donor Registration Forms. It was agreed that these recommendations would be passed on to the Data Advisory Committee for consideration when revising the OPTN data collection forms.
[includes representatives from the Histocompatibility, Kidney/ Pancreas Transplantation, and Minority Affairs Committees]
OPTN. The Subcommittee requested data analyses on two OPOs and on Region 1, each of which has an approved alternative allocation system that eliminates or minimizes HLA points for allocation of deceased donor kidneys (except for zero antigen mismatched mandatory shared kidneys). The following descriptive data for these entities were provided, along with the data for the rest of the United States:
According to candidate's initial PRA level and ethnicity, the Kaplan-Meier median waiting time and percentage transplanted of registrations added to the deceased donor kidney waiting list for first transplant since implementation of the alternative system
Summary of the size of the deceased donor kidney waiting list for first transplant at the beginning and end of the analysis period, and distribution of registrations by ethnicity
According to recipient's most recent PRA level and ethnicity, the most recent data on the distribution of primary kidney transplants performed since implementation of the alternative system
According to recipien's most recent PRA level and ethnicity, the most recent data on Kaplan-Meier graft and patient survival of primary deceased donor kidney transplants performed since implementation of the alternative system
According to recipient's most recent PRA level and ethnicity, most recent data on rejection within one year of transplant for primary deceased donor kidney transplants performed since implementation of the alternative system
According to ethnicity and HLA match points, for recipients who had primary deceased donor kidney transplants and were relisted due to graft failures, the distribution of PRA level at first transplant and at relist
An additional analysis for the Joint Subcommittee provided a tabulation of "positive crossmatch" and "HLA mismatch unacceptable" refusal reasons by PRA, and race/ethnicity for candidates waiting for first and second kidney transplants. Finally, OPTN staff also provided a tabulation of recipient blood type by donor blood type for zero-mismatched transplants and transplants for recipients with PRA>30% and PRA>80%.
SRTR. The requests of the Joint Subcommittee on Kidney Allocation over the past year have focused on examining the effect of HLA and PRA on access to transplantation, survival on the waiting list, and posttransplant survival. The committee examined the impact of eliminating the priority given to matches at the HLA B or HLA B and DR loci on access to kidney transplantation and graft failure. The first analysis looked at whether there were differences in 1) access to transplantation, 2) patient survival, and 3) graft survival for patients in Region 1, which currently uses modified HLA points for deceased donor kidney allocation versus the rest of the nation. Relative deceased donor kidney transplantation rates and two-year graft and patient survival probabilities were compared pre- and post-policy change (1997) for Region 1 and the nation by race and ethnic group. Results showed that in Region 1, removal of B-DR points was associated with 7% more organs transplanted to minority candidates, a small increase in the average mismatch score, and little change in patient survival or graft loss. A related analysis looked at whether OPO and HLA independently affect survival on the deceased donor kidney waiting list. Results showed that death rates on the waiting list differ significantly by OPO and HLA DR antigen. There were no significant differences in death on the waiting list by HLA A and B antigens.
The committee also explored the effect of sensitization (PRA) on access to kidney transplantation and graft failure. The first PRA analysis examined the influence of PRA (baseline, most recent, and peak) on the likelihood of receiving a deceased donor transplant by race, gender, and ethnic group. Results showed that baseline PRA has a bigger effect on transplant rates for AfricanAmericans than for whites. Baseline PRA has a similar effect on access to transplantation for males and females as well as for Hispanics and non-Hispanics. Peak PRA differed in that whites have lower transplantation rates than do AfricanAmericans in the PRA range of 10%-40%. Peak and most recent PRA differ in that females with PRA of 80% or higher have lower transplantation rates than do males.
The second analysis examined recipient sensitization at time of repeat transplant. Results showed that PRA, HLA mismatch at first transplant, and race/ethnicity were significantly associated with receiving a second transplant. PRA and race/ethnicity were also associated with the risk of graft failure for the second transplant, but HLA mismatch at the first transplant was not.Finally, the committee asked the SRTR to investigate factors leading to sensitization at relisting. When looking specifically at factors that increase PRA between the first transplant the time of the second waiting list, it was observed that mismatch at the first transplant was an important predictor, as well as age, sex, race/ethnicity, time since failure of first transplant, and length of survival of the first transplant.
OPTN. OPTN staff provided the Kidney/Pancreas (K/P) Committee with the distribution of pancreata recovered by OPTN/UNOS region and by whether the organ was transplanted locally, regionally, or nationally. This was provided for both pancreas-alone transplants and kidney-pancreas. Additionally, the Committee requested the Kaplan-Meier graft survival of pancreas alone transplants by HLA mismatch level, as well as separate analyses of various patient characteristics for patients who have registered on multiple hospital waiting lists simultaneously and those who have not.
SRTR. The K/P Committee asked for an examination of the impact of HLA mismatchlevel on graft survival for pancreas-alone and pancreas after kidney (PAK) transplants. The Committee was considering removal of priority for HLA matching in pancreas allocation and was interested in any associated increase in the risk of graft failure. The first analysis, examining the impact of HLA mismatch on graft survival for pancreas-alone transplants, found that the relative risk of graft failure for different levels of HLA mismatch (0mm, 1mm, 2mm) differed significantly at the B locus and did not differ significantly at the A and DR loci. Results also showed no significant interaction between mismatch level and time or between mismatch level and year of transplant.
The second analysis examined the impact of HLA mismatch on graft survival for PAK transplants. Results showed that while the adjusted relative risk of graft failure differed significantly by level of HLA mismatch at the B locus for pancreas-alone recipients, there was no significant difference for PAK recipients. In addition, the relative risk of graft failure did not differ significantly by level of HLA mismatch at the A or DR loci for PAK recipients. The results for the B-locus in the pancreas-only model would not be significant if adjusted for the multiple comparisons at the three loci. Since the trend was not replicated with the PAK data, the pancreas-alone original observation may have been due to random chance. These analyses supported the K/P Committee statement for public comment regarding the proposed modification of the pancreas allocation policy by removing priority for HLA matching (with the exception of zero antigen mismatches).
The Committee also requested an investigation of the association between multiple waiting lists in different OPOs and differences in waiting time, waiting list mortality, and/or posttransplant survival for specific patient subgroups. Please see the Patient Affairs heading for a description of this analysis. The K/P Committee also examined the OPO effect on multiple listing. Adjusting for OPO effect does not change the results for waiting time, waiting list mortality, or posttransplant survival. As the ratio of OPO waiting list size to deceased donor kidneys recovered in the OPO increases, the probability of multiple listing increases significantly.
See also Joint Subcommittee on Kidney Allocation
OPTN. Before the inception of MELD, OPTN staff provided the first statistical validation of the MELD system as applied to liver transplant candidates using data voluntarily provided by the members. The initial validation was based primarily on Status 2B patients. For this analysis, the area under the receiver operating characteristic curve was 0.8, indicating that there was an excellent concordance between MELD and 3-month waiting list mortality. Subsequent analyses showed similar results. Since the inception of MELD on February 27, 2002, OPTN staff have provided extensive descriptive data on the MELD-based allocation of deceased donor livers. These data include mean, median, and range of MELD scores overall and by OPTN/UNOS region for waiting patients and for patients removed from the waiting list for transplant, death, and other reasons. Among those removed from the waiting list, there was very little variation in average MELD scores across OPTN/UNOS regions. In addition, MELD data on the number of patients listed and transplanted for hepatocellular carcinoma and exceptions data compiled from regional review board activities was provided.
In support of efforts to determine the feasibility of a split-liver policy, the OPTN provided historical data on the number of times a liver that was a potential candidate for splitting could have been offered to a pediatric patient in an OPTN/UNOS region in which a Status 1 patient was not actively waiting for a transplant. A total of 292 infants on the waiting list of 39 OPOs were on the waiting list at some time during 2001 at the same time that an ABO-compatible liver was available in a region in which no Status 1 candidates were waiting at the time. The number of infants ranged from 1 to 31 at the 39 OPOs. In a related analysis, the OPTN provided data on the number of reduced liver transplants performed in the United States since 1988. A total of 1,278 reduced liver transplants have been performed between 1988 and 2001.
Other general analyses provided an assessment of the completeness of items (pertinent to liver) collected on the Transplant Candidate Registration Form, Transplant Recipient Registration Form, and Transplant Recipient Follow-up Form; an assessment of compliance rates for liver transplant centers in completing these forms; and an analysis of the rate at which liver transplant recipients are lost to follow-up overall and at individual transplant centers. The percentage of liver transplant programs with one-year compliance rates of 100% for forms expected in 2000 varied by form and ranged from 53% to 72%. For the period from October 1, 1987 to December 31, 1999, the overall lost to follow-up rate for liver transplants was 6.6%. Among transplant programs, 112 of 131 (85%) had 10% or fewer liver transplants reported as lost to follow-up.
SRTR. The Liver Simulated Allocation Model (LSAM) has been under extensive development. A working model to assess the impact of changes in allocation rules, such as MELD-based versus status-based allocation and regional sharing versus no regional sharing has been discussed with the L/I Committee. Since its original release in October 2001, the model has been enhanced to better simulate the probability of organ acceptance. Expected posttransplant outcomes have also been improved to better reflect reality. Cox regression models based on empirical patient and donor characteristic data have been developed to serve as the underlying framework for the LSAM. This model will serve as a useful tool for evaluating the potential impact of allocation policy changes, such as regional sharing by MELD.
The SRTR has begun monitoring 30-day outcomes for patients under the new allocation system by ranges of MELD/PELD, and will continue to monitor these outcomes. Transplant versus waiting list mortality by MELD/PELD score will be assessed using time-dependent Cox regression, once six-month posttransplant follow-up data are available for patients receiving a liver under the MELD system. This will facilitate the determination of whether there is a MELD score above which transplantation is futile, and whether patients with MELDs below a particular score should be transplanted or even listed. Additionally, the effects of the change in MELD/PELD over time will be considered and modeled for possible inclusion in the MELD calculation.
The SRTR has also supported the L/I Committee's interest in deceased donor split-liver transplants and how their outcomes compare to whole-liver transplants in both pediatric and adult recipient populations, because more splitting of deceased donor livers would increase the donor pool. Initial results suggest that recipients of deceased donor split livers have comparatively worse survival than whole-liver recipients, and that the benefit for pediatric recipients, compared to remaining on the waiting list, may not be worth the (life-year) cost of splitting to adult recipients.
OPTN. To support the ongoing goal of the MPSC to monitor transplant programs, OPTN staff provides updated reports for the Committee's review at each MPSC meeting. These reports are designed to help identify if there are kidney, kidney-pancreas, pancreas, liver, heart, or lung transplant programs that meet either of the following criteria:
Have waiting list registrations but have not performed any recent transplants
Have active program statuses inconsistent with either waiting list registrations or performed transplants
SRTR. SRTR provides the Data Subcommittee of the MPSC with quarterly reports of risk-adjusted center-specific mortality using the statistical models developed by the SRTR for the Center-Specific Reports. The Data Subcommittee uses these reports to help identify transplant programs that should be reviewed because of poor performance. The reports show the actual one-year survival rate and one-year survival adjusted for the characteristics of the patients. The reports flag programs that fall below a cutoff value that is specified by the OPTN bylaws. The subcommittee may watch the program to see if performance improves in the future or it may ask the program for more detailed information about the characteristics and outcomes of its recent transplants. Based on the additional information, the subcommittee may decide that the lower than expected survival is due to the program serving patients who have characteristics that are not or cannot be included in the statistical model. The subcommittee may offer the program a site visit to help them determine how outcomes can be improved. Ultimately, the subcommittee can recommend that the full committee begin the process of declaring the program not to be a member in good standing.
At the request of the subcommittee, the SRTR presented an alternative proposal for how the subcommittee might select programs for review based on performance. The present review criteria select primarily small transplant
programs, many of whom have deviations from expected survival that are large but not statistically significant. The SRTR proposed a method that would flag only programs with a statistically significant deviation from expected survival and that would select large as well as small programs. The review flag generated by the SRTR method is included in the reports for the subcommittee's consideration.
OPTN. The MAC requested descriptive data analyzing the kidney and kidney-pancreas waiting lists (most recent snapshot and new registrations) by PRA and candidate ethnicity. Additionally, the Committee was interested in the number of A2/A2B-to-B deceased-donor kidney transplants by recipient ethnicity from the transplant centers in OPOs that have performed such transplants as part of their local variance, and whether plasmapheresis treatment was given. Finally, OPTN staff provided the MAC with a requested tabulation of deceased-donor kidney transplants by OPO, recipient ethnicity, and use of expanded criteria donors, where expanded criteria donors are defined as donors over 50 years of age, or with hypertension, or with diabetes.
SRTR.This committee was interested in looking at factors affecting the time spent waiting for a deceased-donor kidney transplant by race groups. Deceased-donor transplantation rates were evaluated by race and ethnic group as time from waiting list to transplant. Over 71,000 candidates on the waiting list during 1994-1997 were followed to December 31, 2000, and censored at removal from waiting list or end of study. Three levels of adjustment were used: 1) basic characteristics including age, sex, diagnosis group, blood type, year on waiting list, PRA closest to date of waiting list, dialysis modality, source of payment, previous transplants, and transfusions; 2) Model 1 plus HLA antigens of candidates; and 3) Model 2 plus OPO region of waiting list. The percentage deficits in transplant rates for the groups African American, Asian, and other were compared to whites, using all levels of adjustments. The percentage deficits between Hispanic and non-Hispanic candidates were similarly compared. Results showed vast deficits in transplantation rates for minority groups as compared to whites. However, as more adjustments are made, namely OPO region and HLA antigen, these deficits are greatly reduced.
Additionally, the committee asked the SRTR to explore the impact of HLA mismatch level and the impact of dialysis duration on deceased-donor kidney patient and graft survival by recipient race and ethnicity. Patient and graft survival were analyzed, and tables were produced showing the relative risk of mortality and the relative of risk of graft failure, as well as survival probabilities, by HLA mismatch level (zero to six mismatches) overall and for whites, African American, Asians, and Hispanics. Similar tables were produced by degree of dialysis duration.
See also Joint Subcommittee on Kidney Allocation
OPTN. The OAC has been involved in a variety of different topics over the past year. Most of these discussions have focused on various types of expanded donors and trends in living donation. One of its main interests has been nonheartbeating donors (NHBDs). For that discussion, the OPTN provided data on the number of OPOs recovering and transplant centers transplanting NHBD kidneys and livers. Univariate analyses were also provided on the survival rates of NHBD liver transplants and the delayed graft function of NHBD kidneys. This discussion also included reviewing some early results of the new kidney pumping and resistance data, only collected since October 25, 1999. The OPTN also provided the Committee with data on the number and outcomes of dual kidney transplants, and the number of transplants, by organ, where the donors were reported to be positive for Hepatitis B Surface Antigen.
The living donation discussion involved looking at data on the number and percentage of centers performing living donor kidney and liver transplants each year since 1988, as well as reviewing the number of living donor deaths reported to the OPTN since October 25, 1999.
SRTR. The OAC requested an investigation of donor characteristics that were associated with increased kidney graft failure. It was determined that four donor traits were very important in predicting graft failure: age, hypertension, CVA as cause of death, and serum creatinine. A scoring system was devised based on the relative risk (RR) of graft failure of the four above traits. The committee agreed that a RR >1.7 was a good cutoff point to distinguish "expanded" or "marginal" donors. This work was subsequently presented to the OPTN/UNOS Board of Directors and a secondary listing for patients on the waiting list that would accept an expanded donor kidney was implemented in September 2002. A manuscript on this topic by Port et al. has been accepted for publication by Transplantation.
The Committee requested two additional analyses pertaining to storage methods when transporting kidneys for transplantation. The SRTR examined the risk of delayed graft function and graft failure for recipients of pulsatile perfused ("pumped") versus nonpumped kidneys. The first request dealt with NHBD kidneys. This analysis used data from an OPTN special study that collected extensive data on kidneys taken from NHBDs. Outcomes for pumped vs. cold-stored kidneys were compared, as was the resistance on these kidneys. In the end, there was not enough data available to come to any conclusions. The Committee then requested an investigation into differences between pumped and nonpumped kidneys using all deceased-donor kidney transplants (not just those from NHBDs). Pumped deceased-donor kidneys had a 43% significantly lower risk of delayed graft function compared to kidneys that were not pumped in heartbeating donors. There was also a reduction in the risk of delayed graft function for NHBD kidneys, although it was not statistically significant. There was no difference in graft survival between pumped and nonpumped deceased-donor kidneys.
OPTN. The Committee requested that OPTN staff conduct their annual OPO survey during the first week of the year. The survey collects from each OPO the following information based on the previous year: the number of donors; the number of kidneys, livers, hearts, lungs, pancreata, and intestines that were recovered; and the number of those organs that were transplanted. The OPOs also are asked to supply their service area population (both based on CMS designation and CMS agreed upon), the number of donors, and the number of organs recovered from NHBDs. UNOS then prepares reports customized for each OPO based on the results of the survey. Each OPO receives a report that details the aggregate data for the entire United States along with its self-reported figures. These same numbers are reported for the nonheartbeating donors. In addition to the raw numbers, all of this information is reported on a per million population basis. Each OPO also receives all of the above information for itself and its ranking among all other OPOs for each category. These data were provided for the previous year (2001) and based on a four-year cycle (1998-2001). These same data were provided to the OPO Committee in aggregate.
The Committee also requested OPO-specific donor referral compliance rates. Each month, OPOs are required to submit, on an individual hospital basis, the number of referrals, the number of eligible donors (based on a death record review project of the Association of Organ Procurement Organizations), and the number of consents that they received on the eligible donors. The Committee requested to view the numbers reported by each OPO and the OPO-specific compliance rates for the submission of this data. Upon review of these data, the OPO Committee recommended a subcommittee be formed to address standards for data submissions and ways to help the OPOs be more compliant.
A third request of the OPO Committee sought to evaluate the required timeframe for OPO submission of refusal organ reasons (known as Potential Transplant Refusal [PTR] data). In order to offer a donor organ to a transplant candidate, the OPO creates a match run list. Each match run is considered closed when the OPO enters a primary refusal code for every candidate whose transplant center declined the offer and the organ type accepted for the recipient, or when efforts to place the organ are stopped without a recipient identified. UNOS policy states that match runs must be closed within 30 days of the run date. Once the match run is closed, the transplant centers offered these organs have 15 days to confirm the refusal code offered by the OPO or to enter a different refusal code. If no offers are made from a particular match run, the OPO closes the list at sequence #0 indicating that no offers were made. The purpose of this analysis was to determine whether 15 days was enough time for the OPO to enter the data. On the basis of this analysis, the OPO Committee recommended that the policy extend the time to 30 days after the match run.
SRTR. The OPO Committee requested an analysis to identify major factors that impact heart recovery by OPOs. The main purpose of the request was to determine why some donor hearts were not being placed. Heart utilization was defined by whether or not the heart was recovered for transplantation. The results showed that lower probability of heart recovery is associated with female donors, African American donors (compared with whites), older donors (age >45), donors who have hepatitis B, and donors whose cause of death was anoxia or cerebrovascular/stroke (compared with head trauma). Higher probability of heart recovery is associated with higher donor body weight, larger fraction of hearts recovered by the OPO in the previous year, and larger average number of calls to transplant centers that the OPO made to place the heart. The results by number of calls may be in part due to many cases with zero calls.
To determine why donor hearts were not being placed, some modifications were made to the previously developed model for heart recovery. The first model showed that higher probability of heart recovery is associated with donor age categories 18-40 (compared with 40-50 and >50), absence of hepatitis B, higher donor body weight, and donors whose cause of death was head trauma (compared with cerebrovascular/stroke). Results for the OPO-level risk factors showed that higher probability of heart recovery is associated with smaller average number of calls to transplant centers that the OPO made to place the heart, larger number of kidney programs, larger number of lung transplants, higher percentage of shared lungs, lower percentage of shared intestines, larger number of heart waiting list candidates, and smaller number of pancreas waiting list candidates. Similar results for donor-level factors and OPO level factors were also shown in the second model. In addition, the second model showed that higher probability of heart recovery is also associated with smaller number of intestine programs, smaller number of lung programs, and larger number of kidney transplants.
In order to identify characteristics of ideal donors, the SRTR performed additional analyses showing that higher probability of placing the heart on the first call or by the second call (comprising more "ideal" donors) is associated with white donors compared with African Americans (perhaps related to prevalence of hypertension), male donors, donors whose cause of death was head trauma (compared with CVA/stroke), donor age category 0-1 compared with 18-40, and age category 18-40 compared with 40-50 and >50, higher donor body weight, and absence of hepatitis B.
OPTN. The OPTN provided, for the most recent three-year period, the following information about patients listed at multiple transplant hospitals:
By organ type, the number, and percentage of patients listed at one, two, and three, or more centers, stratified by candidate age range, gender, blood type, PRA level, race/ethnicity, primary and secondary source of payment, highest education level, and first or repeat transplant
For those patients listed at more than one center, the number transplanted at the first versus subsequent listing centers
Comparable data on patients who were not multiple-listed but changed their center of listing from one center to another
Analyses suggested that compared to patients who are registered on a single hospital waiting lists, multiple-listed patients are more likely to be white, 18-49 years old, privately insured, and with blood group O or B. Multiple-listed kidney candidates are more likely than patients with one listing to have PRA levels greater than 10%.
SRTR.The PAC requested an investigation of the hypothesis that multiple listing is associated with differences in waiting time, waiting list mortality, and/or posttransplant survival for specific patient subgroups. The Committee was considering whether it should recommend disallowing the practice of multiple listing. Time-dependent Cox models showed that the rates of transplantation during periods of multiple listing are two to four times higher than the rates of transplantation during periods of single listing for both kidney and liver patients. Lower rates of waiting list mortality and lower rates of graft failure for multiple-listed kidney patients suggest that kidney patients who are multiple-listed are less sick than kidney patients who are single listed. On the other hand, higher rates of waiting list mortality for multiple-listed liver patients suggest that liver patients who are multiple-listed are sicker than liver patients who are single-listed, although this does not translate into lower posttransplant survival. These analyses supported the PAC statement for public comment to the OPTN committees regarding the policy on multiple listing.
OPTN. The Pediatric Transplantation Committee was interested in the difference between how patients who were awaiting an isolated liver fared compared to patients who were waiting for both a liver and an intestine. The OPTN provided the Committee with data that compared the death rates of pediatric patients in these two groups based on their liver medical urgency status. These data were based on patients ever on the list between 1999-2001. Status 1 patients who were waiting for an isolated liver actually had a significantly higher death rate than those awaiting both a liver and an intestine. For all other medical urgency statuses, patients awaiting both a liver and an intestine transplant had significantly higher death rates than those awaiting an isolated liver.
Additionally, the Committee has a longstanding interest in pediatric donors from whom at least one organ was recovered, but the heart or liver was not recovered. The Committee wanted to know why these organs were not being recovered. The OPTN provided the Committee with extensive information about these donors (recovered over a two-year period) that included demographic, serological, medical, and sociodemographic information. The Committee narrowed the list of over 600 donors down to fewer than 60 about which it wanted more information. After the OPTN provided the Committee with the number of pediatric patients on the list at the time of recovery, the Committee decided it wanted to contact the OPOs that recovered each donor to get more information on why these organs were not recovered. The OPTN is in the process of contacting each OPO for this information.
A third analysis addressed neonatal donors. The Pediatric Committee discussed thoracic allocation policy for in utero and neonatal transplant candidates. The OPTN provided a table showing the number of neonatal organ donors that have been recovered in the United States each year since 1988. In addition to the number of donors, the number of organs recovered and the number of organs transplanted from these neonatal donors were also listed.
Another set of analyses focused on MELD/PELD implementation. As requested, the OPTN provided the Pediatric Committee with an analysis of the liver waiting list, comparing the allocation system then current (as of January 2002) with the MELD/PELD allocation system, slated for implementation on February 27, 2002. The analysis focused on where the first pediatric patient would fall on the list under the two different systems. The Committee wants to watch the new MELD/PELD system closely to ensure that pediatric patients are not disadvantaged by the new system. After viewing this information, the Pediatric Committee requested to see the PELD scores of the pediatric patients who had received a deceased-donor liver transplant under the new system. The tables provided to the Committee displayed the results of the first three full months of transplants under the MELD/PELD system (March 1, 2002 to May 31, 2002). There were 1,226 deceased-donor liver transplants during this period, 126 of which occurred in pediatric recipients (10.3%). Pediatric recipients made up 10.5% of the deceased-donor liver transplants for the year 2001. The data in the tables were based on data as of June 27, 2002. Forty-two (33.3%) pediatric recipients received a transplant as a Status 1 candidate. The remaining 84 pediatric patients received a transplant with a PELD score that ranged from -7 to 37.Of the 1,100 adult patients who received a deceased-donor liver transplant, 79 (7.2%) were Status 1 candidates, 54 (4.9%) were Status 2A candidates, and the remaining 967 received a transplant with a MELD score that ranged from 6 to 40.
Finally, in light of the fact that many centers are now abandoning heart transplantation as the primary treatment option for patients suffering from the hypoplastic left heart syndrome in favor of the Norwood procedure, the Committee requested the number of fetal listings that have actually occurred since 1993 and the outcomes of these listings.
With the advent of UNet on October 25, 1999, an indicator flag was added to in utero listings on the waiting list; before that date, there was no such indication. Since UNet collection began, there have only been six listings indicated as in utero. For listings before UNet, the OPTN identified all listings that occurred prior to the candidate's date of birth, and analyzed those cases to determine if they were indeed in utero listings. The cases that were identified between January 1, 1993, and October 24, 1999, were verified with the transplant centers in which they occurred. The tables provided to the Committee displayed the patient's date of birth, date of listing, diagnosis, and resolution (transplanted, died, too sick to transplant, condition improved). For those patients that were transplanted, the current status (alive, dead, retransplanted) was listed along with the survival time.
SRTR. The SRTR supported the Pediatric Transplantation Committee's interest in evaluating the magnitude of mortality risk for patients with liver tumors while on the waiting list and after transplant compared to children with other diagnoses. The posttransplant analyses indicated that for transplant 1987-1999, patients with a primary diagnosis of malignant neoplasm initially had better survival rates than any other diagnosis. Their survival decreased at six months, and by year two is worse than any other diagnosis. However, relatively few patients have a diagnosis of malignant neoplasm, and their survival experience is quite variable. The results indicate that at five years, the diagnoses divide into two major groups. The survival experience of Group 1 (malignant neoplasm and AHN) was significantly worse than that of Group 2 (all other diagnoses). For more recent liver transplants (1995-1999), the results are consistent but not statistically significant. Splitting the malignant neoplasm group into three subgroups (HC, HBL, Other) showed that patients with HC and Other malignant neoplasm had significantly lower posttransplant survival than the nonmalignant neoplasm groups except for Other/Missing. For more recent liver transplants, the malignant neoplasm patient subgroups became even smaller and patterns were hard to detect. The pretransplant analyses indicated that patients with malignant neoplasm had an 87% higher mortality risk on the waiting list than those without malignant neoplasm. Repeating the analysis splitting the malignant neoplasm group into the same three subgroups, there were no significant differences between groups. The SRTR also looked at survival with or without a transplant for children listed with a diagnosis of liver tumors compared to all other children on the list. Survival was highest for the children without malignant neoplasm, however, this difference was not significant.
The SRTR was also asked to evaluate the degree of inequity in access to transplantation associated with multiple listing. The effect of multiple listing on posttransplant patient and graft survival as well as retransplantation rates was examined. Multiple-listed kidney patients showed a higher risk of mortality after transplant and no statistical difference in risk of graft failure. Multiple-listed liver patients classified as "other" status showed a higher risk of posttransplant mortality. Other liver status categories showed no statistical difference in the risk of posttransplant mortality. For liver patients, the retransplant rate was 3.7 per 100 patient years at risk for single-listed patients and 11.7 for multiple-listed patients. Both multiple- and single-listed kidney patients had retransplant rates of less than 1 per 100 patient years at risk.
The Committee was also interested in determining whether PELD appropriately predicts the risk of mortality for liver/intestine candidates. The results suggest that PELD score is not a significant predictor of waiting list mortality for pediatric liver-intestine patients. These patients had a significantly higher risk of waiting list and posttransplant mortality compared to isolated liver patients. It was concluded that PELD does not appropriately predict the risk of mortality for liver/intestine candidates. To adjust the current PELD score for the increased risk of waiting list mortality among pediatric liver-intestine patients, 12 +/- 8 points would need to be added to the current PELD score.
Comparing waiting list mortality for pediatric patients waiting for a liver transplant vs. a liver-intestine transplant, it was observed that for children under 2 years of age, three-month survival was worse for liver-intestine patients than for liver only patients. There was no difference in three-month survival for older children in either group. The younger children also had lower survival than the older children in each group. Survival at 90 days was unchanged when follow-up was censored at removal from the waiting list due to improved condition.
The SRTR also evaluated the probability of death and transplant on the waiting list at a given MELD/PELD score. The patient population included 17,863 non-Status 1 candidates on the list on February 27, 2002, plus 411 non-Status 1 candidates listed between February 28, 2002, and June 1, 2002. All patients were followed for 30 days from February 27, 2002, or date of listing. The SRTR produced 30-day outcomes tables by ranges of MELD/PELD. Separate reporting was done for HCC patients at exception MELD scores of 24 or 29. These 30-day outcomes tables were also produced by UNOS region and blood type.
OPTN. The two main topics of discussion in the Thoracic Transplantation Committee during the past year have been improving the allocation system through refinement of the match algorithm and increasing the supply of donors. Within its review of the match algorithm, the Committee has focused on a more detailed stratification of heart and lung patients and a broadening of the distribution system. The Committee has reviewed many OPTN analyses of the current heart allocation system to assess whether members are using it appropriately and whether the system adequately classifies patients according to medical urgency. Pre- and posttransplant outcomes by Status 1A criteria and sub-criteria have been provided. The results of this analysis were central to the Committee's decision to modify criterion A for Status 1A. These data have also been essential in the committee's discussion of whether to retain criterion E (i.e., patient does not meet other criteria but has life expectancy of less than seven days). A formal policy proposal has not yet been made, but many discussions have taken place regarding either eliminating it or to include additional restrictions on its use. Additionally, several analyses have been performed to examine the current geographic boundaries in the distribution system. One tabulation provided the number of hearts recovered by each OPO that were transplanted within each of the distribution zones, as well as the import/export ratio.
In addition to improving the allocation system from the transplantation side, the committee has also been extremely interested in improving heart and lung donation. Many of the analyses performed by the OPTN have been related to this topic, including an examination of the consent and recovery process to determine whether organs not currently being recovered that might have been suitable for transplantation. The Committee has become increasingly concerned about organs not recovered due to medical history or serology on which no placement efforts are made, particular in view of the variation in center's acceptance practices. Another analysis regarding donation included the results of an OPO bronchoscopy survey developed by the Committee. As the information gleaned from a bronchoscopy can affect a center's decision to accept a lung offer, the Committee wanted to assess the practice of obtaining bronchoscopies across OPOs.
SRTR. In response to requests from the OPTN Heart Allocation Subcommittee and the OPTN Lung Allocation Subcommittee, the SRTR has performed analyses of waiting list and posttransplant mortality for patients on the waiting list for and who have received hearts and lungs using OPTN/SRTR data supplemented with data from the Social Security Death Master File (SSDMF).
The primary purpose of the OPTN Lung Allocation Subcommittee has been to design a lung allocation system to replace the current waiting-time-based one. The new system will incorporate both waiting list and posttransplant one-year survival. To this end, the Committee has defined four major diagnosis groups for adult patients and one separate group for pediatric patients under 12 years of age. The diagnoses were grouped primarily based on SRTR analyses of waiting list survival probability. The SRTR has simulated possible benefits from using the waiting list model results in actual allocation. The use of waiting list risk-based allocation, as opposed to the current waiting-time-based allocation, could reduce waiting list deaths by 13% to 20% each year-although without incorporation of the posttransplant survival aspect, the posttransplant mortality risk may rise. The SRTR plans to create an allocation system taking into account both waiting list and posttransplant mortality risk, to evaluate this system, and to refine the waiting list and posttransplant models used to calculate these risks.
The primary purpose of the OPTN Heart Allocation Subcommittee has been to evaluate the current heart allocation system. The SRTR has performed a number of analyses to support this goal. The SRTR determined that the differences in heart waiting list mortality for two outlying regions (one with a high risk of mortality, another with a low risk) were not predicted by waiting list size or organ procurement rates. The SRTR identified the status and life support characteristics of heart waiting list candidates that are most closely associated with a benefit from transplantation, including VAD type and brand and other assist devices. These initial analyses showed that heart transplants are almost universally beneficial; in fact, only patients who started in Status 2 ever had transiently elevated posttransplant risk when compared to patients continuing on the waiting list, presumably due to the transplant procedure itself. This risk rapidly dropped below the levels found on the waiting list within one week posttransplant. Finally, the SRTR examined procurement rates among OPOs with and without statewide sharing agreements and concluded that there was no significant association between statewide sharing agreement and heart procurement rates.
SRTR. At the request of this committee, the SRTR provides Part B (Experience Data) of the 2002 OPTN/UNOS Standardized Request for Information (RFI) electronically to transplant centers. These data are now available for download at the SRTR secure web site, www.ustransplant.org. This information is provided by the SRTR as a service to assist centers in completing the RFI, designed by the RFI subcommittee. This is the SRTR's first iteration of the RFI available for download, and plans for future iterations are being discussed and will be determined in the near future.
Transplant centers may log into the secure web site as they do during the review period for Center-Specific Reports and download a PDF file containing Part B of the Standardized RFI. There are a limited number of fields which should be augmented with data from the center, for which the SRTR cannot make a reliable calculation. These data include the readmission rates and also the complication ratios for living donors. The remaining data are provided by the SRTR: graft and patient survival, retransplant rates, multi-organ transplants, transplant volumes, transplantation inpatient length of stay, median waiting time, number of living donors, and average age (for kidney and liver programs). Some of these data, such as the survival calculations, come directly from the Center-Specific Reports, and the remaining data are calculated using similar methodologies.
As part of its contract, the SRTR also provides research and analytic support to the ACOT. The ACOT functions to provide advice to the HHS Secretary on scientific, medical, ethical, and public health issues related to organ procurement, allocation, and transplantation. There are currently eight subcommittees of the ACOT. The full committee meets two to three times per year and all specific research requests are directed to the SRTR for analyses. The summaries below reflect the research discussion and data requests to the SRTR from each of the subcommittees.
The SRTR completed four analyses for this subcommittee.
1) The subcommittee wanted to assess whether the size of the OPO service area population correlates with the candidacy for transplantation and access to transplantation. The SRTR generated tables containing the following statistics: number of kidney registrations per million population, ratio of the number of kidney registrations / number transplants performed in the OPO service area, and median waiting times. There was no apparent trend in the waiting time by OPO size.
2) The subcommittee also examined the benefit of pancreas transplant over remaining on the waiting list for pancreas only. After transplant, pancreas recipients had a higher rate of mortality compared to pancreas candidates on the waiting list, but this difference did not reach statistical significance.
3) The subcommittee asked the SRTR to evaluate the impact of living unrelated (including "altruistic") versus living related kidney donation on graft survival and patient survival posttransplantation. Patients who received a kidney from a living related donor did not have a significantly different rate of graft failure or patient mortality compared to patients who received a kidney from a living unrelated donor.
4) The SRTR studied the effect of transplant center on kidney graft failure. There was not a significant correlation between size of transplant center (measured by number of first deceased donor kidney transplants performed) and relative risk of kidney graft failure. In addition to the SRTR data requests, comments were made about the need for a living donor registry and the committee agreed to consider this as a recommendation.
The SRTR has completed three analyses for this subcommittee. The first involved differences in marginal donor usage (defined by donor age and size) among hospitals of differing sizes. The hospitals that perform more heart transplants do not necessarily use more older (>55) donors or "smaller" donors (e.g., donor weight <70 kg, and recipient height more than 110% of the donor's height). Differing regions use different proportions of older and "smaller" donors, but the regions that use more older donors were not necessarily the same as the regions that use more "smaller" donors.
The second request involved differences in donation rates between organs. The SRTR investigated whether consent is less likely to be obtained for hearts than for other organs. The differences in organ refusal rate are statistically significant, but hearts are in the middle range, with consent withheld most frequently for lungs and intestines and consent being obtained most frequently for kidneys and livers. Emotional reasons were the ones most frequently cited for refusal of consent.
The third request asked the SRTR to evaluate the survival for patients who accepted hearts with ejection fractions less than 50%. To determine if the ejection fraction should be used as a criterion to identify marginal donors of hearts, the SRTR analyzed survival among recipients. While patients receiving hearts from donors with ejection fractions between 30% and 50% had elevated risk (compared with those receiving hearts with a higher ejection fraction), the difference was not statistically significant due to the relatively small numbers of transplants involving ejection fractions less than 50%.
The current analyses requested by this subcommittee include an investigation of reasons for refusal of hearts and an analysis of cumulative inactive time by organ and by current active/inactive status.
This subcommittee focused on identifying specific areas where recommendations for improving liver allocation can be made. It was proposed that guidelines be developed to identify transplant centers prepared to perform living donor liver transplants, along with the development of a set of standards for certification. The SRTR is also working with the subcommittee to explore the possibility of having regional sharing by MELD/PELD ranges and minimal and/or maximal MELD/PELD listing criteria. These questions will be addressed by several analyses requested of the SRTR, including evaluating HLA matching effects on liver transplant outcomes, posttransplant vs. waiting list mortality risk by MELD/PELD to evaluate whether there is a transplant benefit at the lowest and highest scores, and LSAM modeling of regional sharing by MELD ranges.
This subcommittee is interested in public awareness campaigns to increase organ donors. Rather than a national campaign, they would like to target their campaign efforts to specific groups of people. They would like to know who is less likely to donate so they can focus on increasing donation rates for these groups of people. The subcommittee requested that the SRTR compute donor rates by demographics such as age, race, and gender. In addition to the SRTR request, the subcommittee focused on how to evaluate public awareness campaigns. The subcommittee believed that this was a difficult task, but would think of ways to evaluate changed behaviors, selecting variables to analyze.
The subcommittee discussed the issue of brain-dead victims of an assault and how organ donation does not always occur because there are concerns about the investigation into the homicide. The committee discussed ways to get medical examiners to consistently allow the release of the body. Solutions may include legislation, court decisions, education of medical examiners, and asking the Justice Department to undertake a study on this issue. The OPTN data collection forms have medical examiner consent/refusal information. The SRTR studied such counts: In 1998 the percentage of donors blocked by region was 0%-2% except in Region 1, where it was 12%. The subcommittee felt there was substantial underreporting. The subcommittee also discussed organ donor cards and the issue of intent vs. consent when an individual signs a donor card.
The subcommittee also asked the SRTR to examine the number of organs recovered or not recovered for different combinations of donor and family opinions using OPTN/SRTR data about donor wishes and family wishes. The subcommittee was also interested in obtaining information on how many organs were retrieved using the 24-hour period law (when death occurs, if family members are not available, there is a 24-hour waiting period before any organ can be retrieved) and on tissue donors from the eye bank community.
The SRTR was asked to examine the distribution of shared organs and expanded organs by organ type, year, and OPO. The SRTR found significant variation in the percentage of organs shared (out) by OPO. The percentage of shared out organs by OPO was negatively correlated with the number of organs procured in that OPO (transplanted at any OPO). This means that the more organs procured at the OPO, the lower the percentage of organs shared out of the OPO. This was true for each organ type and may reflect in part the OPO rise.
There was significant variation in the proportion of expanded organs by year and there was a significant trend of an increasing age of expanded donors being transplanted by year, with the average increase being 0.5 percentage points per year. Also, there was significant variation in proportion of expanded organs by organ type. Expanded donor kidneys, hearts, and livers are used much more often than other organs. There was a significant trend of an increasing percentage of expanded donors being transplanted by year for hearts, kidneys, and livers. There was no significant time trend for kidney/pancreas, pancreas, lung, heart/lung, or intestines.
There was significant variation in the proportion of expanded organs by OPO and by year. Each OPO's overall percentage of expanded donor organs procured was compared to the overall average of 13%. There was no correlation between the percentage of expanded donor organs transplanted and the overall number of organs transplanted. There were significant differences among the OPOs in terms of the number of donors per million population. An overall average of 21 donors per million population was observed.
The ACOT Multicultural Issues Subcommittee requested two analyses from the SRTR. The first analysis showed the following results: 1) Per 10,000 deaths, rates of deceased donor organ donation are similar for all races. 2) The transplantation rate is lower for African American registrants (vs. whites) on the kidney, pancreas, kidney-pancreas, heart, and lung waiting lists, but higher for African American registrants on the liver waiting list. The transplantation rate is lower for Asian registrants (vs. whites) on the kidney, kidney-pancreas, and liver waiting lists, but higher for Asian registrants on the heart waiting list. The transplantation rate is lower for Hispanic registrants (vs. non-Hispanics) on the kidney and lung waiting lists but higher for Hispanic registrants on the heart waiting lists. 3) The waiting list mortality rate is lower for African American registrants on the kidney waiting list, but higher for African American registrants on the kidney/pancreas, liver, and lung waiting lists. The waiting list mortality rate is lower for Asian registrants on the kidney waiting list, but higher for Asian registrants on the lung waiting list. The waiting list mortality rate is lower for Hispanic registrants on the kidney waiting list, but higher for Hispanic registrants on the lung waiting list. 4) For African American vs. white patients, there are large and significant geographic differences in the waiting list rate (26%-80% lower for African-Americans). Counts of all end-organ failure patients are available only for end-stage renal disease. Similar rates cannot be calculated because the denominator for patients with other organ failures is unavailable.
The second analysis looked at how geography impacts organ allocation via differences in state Medicaid coverage. Results indicate that Medicaid-only coverage is observed more among all minorities than among whites at wait-listing and at transplantation. Additionally, large regional variations are observed.
The Clinical Issues Subcommittee has devoted its attention to several issues related to living donation. The subcommittee has not made any data or analytic requests of the SRTR.
In addition to providing specific, committee-directed analyses as described above, the OPTN and SRTR contractors meet the data needs of other requestors through a variety of mechanisms.
A large part of what UNOS does under the OPTN contract involves collecting, processing, validating, analyzing, and reporting on transplant data. An OPTN goal is to make information contained within the OPTN database available to all interested groups and individuals involved in various aspects of transplantation and medical science. Several departments at UNOS together respond to over 21,000 annual requests for data and information resources from the public as well as the clinical and scientific research communities.
The Communications Department may be the first point of contact for many individuals interested in organ procurement and transplantation information. Roughly, one-fourth of those receiving information from the Communications Department are representatives of the news media and one-sixth are OPTN/UNOS members. Types of information provided include general information about UNOS, the OPTN, and the donation/transplant process (38% of requests) and also basic statistics on the transplant waiting list, donors, and transplant recipients (17% of all requests). Overall, over 3,500 requests are handled solely by this department.
Other individuals needing more patient-oriented information may contact (or be referred to) the Professional Services Department, which provides patient information services to patients, family members, and the public. In the past year, over 14,000 requests for information on transplant patients, data, policy, or donation were completed in this manner. (See Appendix E for more information on these services.)
A final source of data and information is the Research Department, which provides a wealth of readily accessible data for Internet users, supplies specific numbers and generates custom analyses for those with further questions, and prepares comprehensive data analysis or files for approved scientific studies and public-use applications, as described below.
Internet access. A primary way that transplant-related data are made available to the public is through the Internet at www.optn.org. The most frequently requested data are provided for instant access and download in a variety of national, regional, state, or center-level formats. Data include key statistics about patients on the OPTN national waiting list, as well as donors and transplants to date, and are updated as frequently as every week to allow the user to get the most recent data possible. Also, the advanced reporting feature provides key data in a variety of custom report formats that can meet specific needs of individual data users.
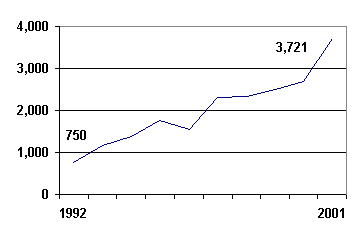
OPTN Data Request System. UNOS receives many additional requests for statistical data. During 2001, UNOS Research staff completed 3,721 data requests, a 28% increase from the 2,688 requests completed in 2000. Appendix Figure H-1shows the increase in completed requests over the past 10 years, and Appendix Table H-1provides the distribution of requests by source.
|
Request Source |
# Requests |
|---|---|
|
OPTN Members Students / Researchers Nonmember Medical Institutions Patients, Donors, Families Private Companies Other |
1,048 833 621 382 371 466 |
|
TOTAL |
3,721 |
Specific requests received at UNOS by phone, fax, or e-mail enter a tracking database and are fulfilled through a number of ways described below.
If the data requested are readily available, they are provided immediately. This includes data found in any of the above-described website reports, the Annual Report, or one of over 100 Standard Reports updated monthly by the Research Department with the most up-to-date data available. Roughly, 60% of all requests can be answered in this manner, without the time necessary for custom report generation.
Requests that require 20 hours or less of programming time are prioritized and assigned to a staff member for completion. Most requests are filled within two to three weeks. Close to 1,000 of these types of programming requests were completed in the past year, and in most cases there was no fee charged for these requests.
Extensive data requests include all nongovernmental research proposals and data requests that are estimated to require more than 20 hours of programming or any statistical analysis. These requests must include a concept paper that provides a purpose for the study, describes and lists the data needed to perform the analysis, and indicates in a general way the type of statistical analysis desired. Concept papers are reviewed by OPTN staff and approved by the Chair of the OPTN/UNOS Data Advisory Committee to ensure that the data are available and that the proposal and methods are clear and appropriate. Guidelines for preparing and submitting a concept paper are available from the Research Department. Upon study approval, Staff are designated to provide programming and/or analytical support for the study and help refine the study guidelines if needed.
In 2001, the OPTN began releasing its Standard Transplant Analysis and Research (STAR) Files. These public-use files contain a comprehensive set of transplant data and allow analysis of more than 500 variables on transplant recipients collected on the OPTN data collection forms. The STAR Files include extensive documentation of each variable, including the proper format and the history of each variable's collection. Staff are also available to researchers to answer any specific questions about the data.
Currently individual files are available with transplant or transplant follow-up data for liver, kidney/pancreas, and thoracic transplant events. Research scientists with statistical programming skills are the primary audience for these files, which allow users to do their own quantitative analyses, test theories, and possibly provide new insights into transplant outcomes. Although individual patients are not identifiable inthese files, a user agreement must be completed when requesting STAR files. Files are free for OPTN/UNOS members, and others are charged a nominal fee.
Data requests can be processed by calling the UNOS Data Request telephone number at 804-782-4876, faxing requests to 804-782-4994, sending an e-mail to datarequest@unos.org, or accessing the OPTN Web site at www.optn.org.
In addition to providing research support to the OPTN, ACOT, and other government entities, the SRTR also provides information to the wider transplant community by making a variety of data sources available and directly responding to data requests for information and analyses.
SRTR Transplant Data Sources. The SRTR produces comprehensive Center-Specific Reports (CSRs) and OPO-Specific Reports (OSRs), which provide a wealth of information to the transplant community. These reports present ongoing analyses of pre- and posttransplant data and are updated twice a year on www.ustransplant.org. Accurate and timely reporting of performance measures for these organizations is useful to the organizations themselves for quality improvement activities, to transplant candidates, and to families of potential donors (See Appendix G for more information on CSRs and OSRs). The SRTR adapts these reports for the specific needs of other interested parties such as the OPTN Transplant Administrators Committee for submission to payers, the Centers for Medicaid and Medicare Services (CMS), and the OPTN Membership and Professional Standards Committee.
The SRTR also disseminates information to the transplant community by creating a comprehensive set of Reference Tables and the corresponding Technical Notes published in the Annual Report. These Reference Tables and Technical Notes are widely used by transplant professionals, researchers, patients, and their families.
SRTR Data Request System. In order to prioritize and manage the volume of all its nongovernmental data requests, the SRTR categorizes the various types of data requests into four broad categories outlined below:
Request for Information (RFI).This category includes general requests that can be answered using existing published sources (i.e., the Annual Report, the Center-Specific Reports) or simple computer tabulations. In general, the SRTR's approach to handling nongovernmental data requests begins with producing comprehensive and well-indexed data sources, such as the Annual Report, so that as many data requests as possible can be answered using these sources.
Request for Analysis (RFA). This type of request generally requires more complex analyses, programming, statistical support, or clinical review. SRTR staff review each RFA to determine the time and resources required to complete it. Depending on the complexity of the request, an RFA may be assessed a nominal hourly fee.
Standard Analysis Files (SAF). To make transplant data accessible to other researchers, the SRTR releases standard data files with documentation and technical support from SRTR staff. The SAFs allow researchers to conduct their own analyses of SRTR and Post-Transplant Tumor Registry (PTTR) data. The SAFs contain a standardized set of data fields without patient identifiers on a CD-ROM, which a requestor can obtain after a Data Use Agreement has been signed. The SAFs are available to the general transplant community for a nominal fixed fee. In the event that that there is a request for a SAF with patient identifiers, a different fee is assessed since more time and effort is needed to process the request, obtain approval from the SRTR Scientific Advisory Committee (SAC) and the SRTR Project Officer (PO), prepare the files, and provide technical support.
Custom Data Files (CDF). In addition to standard data files, the SRTR also provides customized data files. These customized files are similar to SAFs in that they provide data to researchers so that they can perform their own analyses. Typically, a CDF requires additional time and effort to create the specially tailored files and documentation, as well as more technical support from SRTR staff. Depending on the complexity of the customized file, a nominal hourly fee is assessed, which represents the time and effort required to process the request, obtain SAC and PO approval if patient identifiers are requested, create the customized file and documentation, distribute the files and provide follow-up technical support.
Within each category, requests generally are filled in the order, in which they are received, with some exceptions. For instance, if a number of closely related requests are received, then they may be combined and filled as a group. Furthermore, the calendar time estimate for completing any nongovernmental data request is contingent on the time commitments allocated to other existing requests. The SRTR's objectives in prioritizing nongovernmental data requests is to efficiently disseminate information to the wider transplant community and to establish processes to manage and monitor the volume of data requests. Since award of the SRTR contract in October 2000, URREA has received over 1,000 different data requests. Appendix Table H-2 shows the distribution of requests by requestor category and Appendix Figure H-2 illustrates the substantial increase in volume of nongovernmental data requests since October 2001.
|
Requestor Category |
# Requests (October 2001 - September 2002) |
|---|---|
|
Transplant centers Patients Medical professionals Organ procurement organizations Private companies Students Researchers Media Other |
230 153 73 29 41 29 22 14 284 |
|
TOTAL |
875 |
Requests for information, data, and statistical analyses can be processed by calling the SRTR at 1-734-665-4108, ext. 267, emailing mail@ustransplant.org, or accessing the SRTR web site at www.ustransplant.org.
Below is a reference listing of peer-reviewed abstracts and publications on which OPTN or SRTR staff served as authors during October 2001 through October 2002. For assistance locating an abstract or publication, please contact the OPTN or SRTR as listed.
1. Bennett LE, Babcock WD, Kauffman HM. Does Having a Thoracic Transplant Program Affect OPO Heart and Lung Donation Rates? Am J Transplant 2002, 2(Suppl 3):A525.
2. Bresnahan BA, Johnson CP, Cherikh WS, Rosendale J, Hariharan S. Improved Long-Term Kidney Survival: Impact of Zero Mismatch and Preemptive Transplantation: An Analysis of UNOS Data. Am J Transplant 2002, 2(Suppl 3):A558.
3. Callender CO, Cherikh WS. The Impact of Donor-Recipient Ethnicity Combinations on Cadaveric Kidney Graft Survival: A Multivariate Analysis. Am J Transplant, 2002, 2(Suppl 3):A564.
4. Callender CO, Cherikh WS. Kidney Graft Survival Affected by Donor-Recipient Ethnicity? Transplantation 2002, 74(4):A3399.
5. Cherikh WS, Kauffman HM, Maghirang J, Bleyer AJ, Johnson CP. Efficacy Comparison of Discharge Immunosuppressive Regimens in Cadaveric Kidney Transplantation: What is the Best Combination? Am J Transplant 2002, 2(Suppl 3):A1309.
6. Cherikh WS, Ting A. Decreased Access to Renal Transplants in African Americans: Is Sensitization an Issue? Am J Transplant 2002, 2(Suppl 3):A600.
7. Cherikh WS, Ellison MD, Callender CO. Trends in Living Donor Kidney Transplants: Do Minorities Benefit? Transplantation 2002, 74(4):A1640.
8. Cherikh WS, Kauffman HM, Bleyer AJ. Donor Smoking History Associated with Increased Graft Loss in Cadaveric Kidney Transplantation. Transplantation 2002, 74(4):A2223.
9. Edwards E, Harper A, Freeman R. MELD Validation by Medical Urgency Status. Am J Transplant 2002, 2(Suppl 3):A646.
10. Edwards E, Harper A, Freeman R. MELD Validation by Medical Urgency Status. Liver Transpl 2002, 8(6):A68.
11. Egan TM, Bennett LE, Garrity ER, Grover FL, Ring S, Robbins RC, Trulock EP, Wood ED. Are there Predictors of Death at the Time of Listing for Lung Transplant? J Heart Lung Transplant 2002, 21(1):A275.
12. Ellison MD, McBride MA, Taranto SE, Delmonico FL, Kauffman HM. Living Renal Donation: When Transplant Donors Become Transplant Candidates. Am J Transplant 2002, 2(Suppl 3):A351.
13. Fischer S, Bennett LE, Strueber M, Waddell TK, Niedermeier J, Keshavjee S, Haverick A. Outcome Following Simultaneous and Sequential Lung-Liver Transplantation: Analysis of the ISHLT/UNOS Joint Thoracic Registry. J Heart Lung Transplant 2002, 21(1):A149.
14. Freeman R, Harper A, Edwards E. There is Minimal Variation Among OPTN/UNOS regions in Prospectively Collected MELD Scores for Patients Receiving Liver Transplants. Am J Transplant 2002, 2(Suppl 3):A453.
15. Freeman R, Harper A, Edwards E. There is Minimal Variation Among OPTN/UNOS regions in Prospectively Collected MELD Scores for Patients Receiving Liver Transplants. Liver Transpl 2002, 8(6):A254.
16. Goldfarb-Rumyantzev AS, Maghirang J, Scandling J. Donor and Recipient Body Mass Index and Three Year Cadaveric Kidney Allograft Outcome. Am J Transplant 2002, 2(Suppl 3):A148.
17. Goldfarb-Rumyantzev AS, Scandling J. Maghirang J, Smout RJ, Horn S. Predicting the Three Year Allograft Survival in Patients with Cadaveric Renal Transplant Using Logistic Regression (LR) Model. Am J Transplant 2002, 2(Suppl 3):A567.
18. Goldfarb-Rumyantzev AS, Maghirang J, Scandling J. Prediction of the Three Year Allograft Survival in Patients with Cadaveric Renal Transplant Using Tree-Based Model. Am J Transplant 2002, 2(Suppl 3):A1113.
19. Higgins RSD, Bennett LE, DeAnda A, Kasirajan V. Retrospective Assessment of Echocardiography as a Screening Tool in Cadaveric Heart Donors. Am J Transplant 2002, 2(Suppl 3):A526.
20. Kauffman HM, McBride MA, Cherikh WS, Spain PC, Marks WH, Roza AM. UNOS Transplant Tumor Registry Incidence of Donor Related Malignancies. Am J Transplant 2002, 2(Suppl 3):A902.
21. Marino G, Rustgi VK, Halpern MT, Lu AD, Tolleris CB. Role of Gender Mismatch and Graft Failure in Patients Undergoing Liver Transplantation. Hepatology 2002.
22. Meyer DM, Bennett LE, Torres F, Jessen ME, Novick RJ. Impact of Recipient Age and Procedure Type on Survival Following Lung Transplantation for Interstitial Pulmonary Fibrosis. J Heart Lung Transplant 2002, 21(1):A245.
23. Perez RV, Ormond DB, Rosendale JD, Gandhi MM, Gallay BJ, McVicar JP, Troppmann C. Early Renal Allograft Function After Laparoscopic vs. Open Live Donor Nephrectomy: Analysis of the UNOS National Database. Am J Transplant 2002, 2(Suppl 3):A113.
24. Rosendale JD, Kauffman HM, McBride MA, Chabalewski FL, Zarrof JG, Garrity ER, Delmonico FL, Rosengard BR. Aggressive Pharmacological Donor Management Results in More Transplanted Organs. Am J Transplant 2002, 2(Suppl 3):A943.
25. Rosendale JD, Kauffman HM, McBride MA, Chabalewski FL, Zarrof JG, Garrity ER, Delmonico FL, Rosengard BR. Hormonal Resuscitation Yields More Transplanted Hearts With Improved Early Function. Transplantation 2002, 74(4):A608.
26. Rustgi VK, Marino G, Halpern MT, Tolleris CB. Impact of Center Volume on Liver Transplant Outcomes. Hepatology 2002.
27. Rustgi VK, Marino G, Halpern MT, Poltkin JS, Tolleris CB. Impact of Body Mass Index (BMI) on Graft Failure and Overall Survival Following Liver Transplant: Hepatology 2002.
28. Rustgi VK, Marino G, Halpern MT, Poltkin JS, Tolleris CB. Impact of Donor Age on Graft Survival among Liver Transplant Recipients. Analysis of the UNOS Database: Hepatology 2002.
29. Siddiqi N, Johnson CP, McBride MA, Maghirang J, Bresnahan BA. Correlation Between Risk Factors for Renal Dysfunction (serum creatinine > 1.5 mg/dl or 2.0 mg/dl) at 1 Year Post-Transplant and Long-Term Graft Failure: An Analysis of UNOS Data. Am J Transplant 2002, 2(Suppl 3):A1312.
30. Takemoto SK, Harper A, Tolleris CB, Klohe E, Pollack MS, Leichtman A, Ting A, Ellison MD. Two Year Analysis of the UNOS CREG Allocation Variance. Am J Transplant 2002, 2(Suppl 3):A40.
31. Takemoto SK, Tolleris CB, Klohe E, Pollack MS, Leichtman A, Ting A, Ellison MD. Two-Year Analysis of the UNOS CREG Allocation Variance. Hum Immunol 2001, 62(Suppl 1):S34.
32. Ting A, Cherikh WS. Pretransplant Sensitization: Is it a Cause for Decreased Access to Cadaveric Renal Transplants in African Americans? Transplantation 2002, 74(4):A1093.
33. Ting A, Edwards LB. Distribution of Kidney Transplants from Cadaveric Donors in Different Racial Groups Using HLA-A,B,DR for Allocation. Human Immunol 2002, 63(Suppl 1):S65.
34. Tolleris CB, Ting A. HLA Typing for Non-Renal Organ Transplantation: Is It Done, Is It Necessary? Hum Immunol 2002, 63(Suppl 1):S70.
35. Vaidya S, Cherikh WS, Ting A, Distant DA. Analysis of HLA "Blanks" in Various Ethnic Groups of Patients on the OPTN/UNOS Cadaveric Kidney Waiting List. Hum Immunol 2001, 62(Suppl 1):S96.
36. Vaidya S, Cherikh WS, Ting A, Distant D. Analysis of HLA Blanks in Various Ethnic Groups of Patients and Cadaveric Kidney Donors on the UNOS/OPTN Waiting List. Transplantation 2002, 74(4):A3042.
1. Bennett L, Keck B, Hertz M, Trulock E, Taylor D. Worldwide Thoracic Organ Transplantation: A Report from the UNOS/ISHLT International Registry for Thoracic Organ Transplantation. In: Cecka J, Terasaki P (eds). Clinical Transplants 2001. Los Angeles:UCLA Tissue Typing Laboratory 2002:25-40.
2. Boucek MM, Edwards LB, Keck BM, Trulock EP, Taylor DO, Mohacsi PJ, Hertz MI. The Registry of the International Society for Heart and Lung Transplantation: Fifth Office Pediatric Report 2001 to 2002. J Heart Lung Transplant 2002, 21(Suppl 8):827-840.
3. Chabalewski F, Ellis JM. A comparative study of 1993 and 2000 organ donation and transplantation-related content in undergraduate nursing schools. Prog Transplant. In press.
4. Chabalewski F, Seem D. The national transplantation system. In: Cupples S, Ohler L (eds.) Nursing Secrets in Transplantation. Philadelphia: Hanley & Belfus Medical Publishers 2002. In press.
5. Edwards C, Chabalewski FL. The organ and tissue donor. In: McQuillan KA, Von rueden KT, Hartsock RL, Flynn MB, Whalen E (eds.) Trauma Nursing: From Resuscitation through Rehabilitation 3rd ed. Philadelphia: WB Sanders Co. 2002.
6. Edwards E, Harper A. Application of a Continuous Disease Severity Score to the OPTN Liver Waiting List. In: Cecka J, Terasaki P (eds). Clinical Transplants 2001. Los Angeles:UCLA Tissue Typing Laboratory 2002:19-24.
7. Ellison MD, McBride MA, Taranto SE, Delmonico FL, Kauffman HM. Living Kidney Donors in need of Kidney Transplants: A Report from the OPTN. Transplantation. In press.
8. Feng S, Buell JF, Cherikh WS, Deng MC, Hanto DW, Kauffman HM, Leichtman AB, Lorber MI, Maters RG, McBride MA, Metzger RA, Nolte FS, O'Connor KJ, Roth D, Terrault NA, Henry ML. Organ Donors with Positive Viral Serology or Malignancy: Risk of Disease Transmission by Transplantation. Transplantation. In press.
9. Hariharan S, McBride MA, Cherikh WS, Tolleris CB, Bresnahan BA, Johnson CP. Post-Transplant Renal Function in First Year Predicts Long-Term Kidney Transplant Survival. Kidney International 2002, 62(1):311-318.
10. Harper A, Edwards E, Ellison M. The OPTN Waiting List, 1988-2000. In: Cecka J, Terasaki P (eds). Clinical Transplants 2001. Los Angeles:UCLA Tissue Typing Laboratory 2002:73-85.
11. Hertz M, Taylor D, Boucek M, Mohacsi P, Edwards L, Keck B. The Registry of the International Society for Heart and Lung Transplantation: Nineteenth Official Report - 2002. J Heart Lung Transplant 2002, 21(9): 950-970.
12. Kasiske BL, Snyder MS, Matas A, Bennett LE, Ellison MD, Gill JS, Kausz AT. Preemptive Kidney Transplantation: The Advantage and the Advantaged. J Am Soc Nephrol. In press.
13. Kauffman HM, Cherikh WS, Cheng Y. Posttransplant Malignancies: A Problem, a Challenge, and an Opportunity (Editorial). Liver Transpl 2002, 8(5):488-490.
14. Kauffman HM, McBride MA, Cherikh WS, Spain PC, Delmonico FL. TransplantTumor Registry: Donors with Central Nervous System Tumors. Transplantation 2002, 73(4):579-582.
15. Kauffman HM, McBride MA, Cherikh WS, Spain PC, Hanto DW, Delmonico FL. Donor Related Malignancies. Transplant Rev. In press.
16. Kauffman HM, McBride MA, Cherikh WS, Spain PC, Marks WH, Roza AM. Transplant Tumor Registry: Donor Related Malignancies. Transplantation 2002, 74(3):358-362.
17. Mahoney R, Taranto S, Edwards E. B-Cell Crossmatching and Kidney Allograft Outcome in 9031 United States Transplant Recipients. Hum Immunol 2002, 63:324-335.
18. Mange KC, Cherikh WS, Maghirang J, Bloom RD. A Comparison of the Survival of Shipped and Locally Transplanted Cadaveric Renal Allografts. N Engl J Med 2001, 345(17):1237-1242.
19. Rosendale JD, Chabalewski FL, McBride MA, Garrity ER, Rosengard BR, Delmonico FL, Kauffman HM. Increased Transplanted Organs from the Use of a Standardized Donor Management Protocol. Am J Transplant 2002, 8(2):761-768.
20. Rosendale JD, Kauffman HM, McBride MA, Chabalewski FL, Zarrof JG, Garrity ER, Delmonico FL, Rosengard BR. Aggressive Pharmacological Donor Management Results in More Transplanted Organs. Transplantation. In press.
21. Rosendale J, McBride M, Davies D. Organ Donation in the United States: 1988-2000. In: Cecka J, Terasaki P (eds). Clinical Transplants 2001. Los Angeles:UCLA Tissue Typing Laboratory 2002:87-96.
22. Rustgi V, Marino G, Halpern MT, Johnson LB, Umana WO, Tolleris CB. Role of Gender and Race Mismatch and Graft Failure in Patients Undergoing Liver Transplantation. Liver Transpl 2002, 8(6):514-518.
23. Shorr AF, Davies DB, Nathan SD. Outcomes for Patients with Sarcoidosis Awaiting Lung Transplantation. Chest 2002, 122(1):233-238.
24. Stegall MD, Dean PG, McBride MA, Wynn JJ. The Advantages of Sharing Cadaveric Kidneys in the Zero Mismatch Era. Transplantation 2002, 74(5):670-675.
25. Ting A, Bennett LE. HLA in the Allocation of Kidneys from Cadaveric Donors in the United States. Transplantation. In press.
26. Waddell TK, Bennett LE, Kennedy R, Todd TRJ, Keshavjee SH. Heart-Lung or Lung Transplantation for Eisenmenger's Syndrome. J Heart Lung Transplant 2002, 21(7):731-737.
27. Zaroff JG, Rosengard BR, Armstrong WF, Babcock WD, D'Allesandro A, Dec W, Edwards NM, Higgins RD, Jeevanandam V, Kauffman M, Kirklin JK, Large SR, Marelli D, Peterson TS, Ring WS, Robbins RC, Russell SD, Taylor DO, Van Bakel A, Wallwork J, Young JB. Consensus Conference Report: Maximizing Use of Organs Recovered from the Cadaver Donor: Cardiac Recommendations 2002. Circulation 2002, 106 (Suppl 7):836-41.
1. Burdick RA, Leichtman AB, Bragg JL, Cibrik DM, Dykstra DM, Port FK. Obesity is Associated with Increased Risk of Rejection, Delayed Graft Function, Graft Loss and Patient Survival. Am J Transplant 2002, 2(Suppl 3):A966.
2. Distant DA, Port FK, Wolfe RA, Ashby VB, Rush SH, Wynn JJ, Roberts JP, Held PJ. Are HLA and Geographic Regions Associated with Kidney Transplantation Rates among Waitlisted US Candidates by Minority Group? Am J Transplant 2002, 2(Suppl 3):A500.
3. Dykstra DM, Gaylin DS, Carroll CE, Shapiro JR, Beronja N, Rubin, RJ, Young EW, Held PJ. Patient Access to Kidney Transplantation: Results from ESRD Managed Care Demonstration (Demo). Am J Transplant 2002. 2 (Suppl 3):A497.
4. Freeman RB, Wiesner RH, Wolfe RA, Rush SH, Dykstra DM, Port FK, Merion, RM. Analysis of Meld Mortality Risk Data from Waitlisted Liver Candidates in the US. Am J Transplant 2002, 2(Suppl 3):A454.
5. Kayler LK, Rasmussen CS, Maraschio MA, Cibrik D, Punch JD, Campbell DA, Leichtman AB, Magee JC, Rudich SM, Arenas JD, Ojo AO, Merion RM. Gender Imbalance in Living Donor Renal Transplantation: Analyses of Data from the Scientific Registry of Transplant Recipients (SRTR). Am J Transplant 2002, 2(Suppl 3):A353.
6. Kayler LK, Rasmussen CS, Punch JD, Rudich SM, Magee JC, Maraschio MA, Arenas JD, Campbell DA, Merion RM. Results of Liver Transplantation in Children with Metabolic Disorders. Am J Transplant 2002, 2(Suppl 3):A857.
7. Leichtman AB, Bragg JL, Hulbert-Shearon TE, Held PJ, Gillespie BW, Port FK, Ojo AO, Merion RM, Wolfe RA. A Predictive Model of the Probability of Kidney Waitlist Outcomes. Am J Transplant 2002, 2(Suppl 3):A34.
8. Leichtman AB, Bragg JL, Hulbert-Shearon TE, Held PJ, Newmann J, Gillespie BW, Port FK, Ojo AO, Merion RM, Wolfe RA. A Predictive Model of Cadaveric Kidney Transplant Waitlist Outcomes. Transplantation 2002, 74(4):363.
9. Maraschio MA, Burdick RA, Dykstra DM, Punch JD, Wolfe RA, Port FK, Merion RM. Survival Benefit for Obese Recipients of Cadaveric Renal Transplants Compared to Obese Patients Awaiting Transplantation. Transplantation 2002, 74(4):217-218.
10. Merion RM, Ashby VB, Wolfe RA, Hulbert-Shearon TE, Metzger RA, Ojo AO, Held PJ, Port FK. Mortality Risk for Expanded Donor Kidney Recipients Compared With Waitlisted Dialysis Patients. J Am Soc Nephrol 2002, 13:47A-48A.
11. Merion RM, Guidinger MK, Newmann JM, Ellison MD, Port FK, Wolfe RA. Prevalence and Outcomes of Multiple Listing in Cadaveric Renal Transplantation. J Am Soc Nephrol 2002, 13:189A.
12. Merion RM, Murray S, McCullough KP, Bustami RT, Held PJ, Port FP, Dickinson DM, Egan TM, Grover FL. Modeling of Cadaveric Lung Allocation Based on Waitlist Mortality Risk. Am J Transplant 2002, 2(Suppl 3):A523.
13. Merion RM, Wolfe RA, Dykstra DM, Leichtman AB, Gillespie BW, Held PJ. Longitudinal Assessment of Mortality Risk among Waitlisted Liver Transplant Candidates. Am J Transplant 2002, 2(Suppl 3):A457.
14. Merion RM, Wolfe RA, Dykstra DM, Moskal KS, Roberts JP, Freeman RB, Held PJ. Computer Simulation Predictions of the Impact of Meld-Based Allocation on Liver Waiting List Mortality. Am J Transplant 2002, 2(Suppl 3):A458.
15. Murray S, Merion RM, McCullough KP, Bustami RT, Gillespie BW, Wolfe RA, Grover FL, Egan TM. Diagnosis-Based Models of Lung Transplant Waiting List Mortality. Am J Transplant 2002, 2(Suppl 3):A524.
16. Ojo AO, Sablinski T, Roberts JP, Wolfe RA, Leictman AB, Merion RM, Schmouder RL, Stolk J. Prospectively Validated Scoring Index to Predict Delayed Graft Function (DGF) in Cadaveric Renal Transplantation.Am J Transplant 2002, 2(Suppl 3):A547.
17. Ojo AO, Webb RL, Arndorfer JA, Leichtman AB, Port FK, Merion RM. Kidney Failure in Recipients of Liver, Heart, Lung, and Intestinal Transplantation. Am J Transplant 2002, 2(Suppl 3):A1307.
18. Port FK, Bragg JL, Metzger RA, Wolfe RA, Wynn JJ, Delmonico FL, Gillespie BW, Dykstra DM, Merion RM, Held PJ. Who Receives Expanded Criteria Donor Kidneys and what is the Early Versus Late Outcome? Am J Transplant 2002, 2(Suppl 3):A147.
19. Port FK, Bustami RT, Wolfe RA, Leichtman AB,Merion RM, Ojo AO. Immunosuppression and the Risk of Post Transplant Malignancy Among Cadaveric Kidney Transplant Recipients. J Am Soc Nephrol 2002, 13:369A.
20. Roberts JP, Port FK, Wolfe RA, Rush SH, Bragg JL, Ashby VB, Distant DA, Wynn JJ, Held PJ. Eliminating Points For HLA B Matches Benefits Access to Kidney Transplantation for Minorities with Minimally Increased Risk of Graft Loss. Am J Transplant 2002, 2(Suppl 3):A32.
21. Silas P, Ojo AO, Webb RL, Arndorfer JA, Leichtman AB, Wolfe RA, Cibrik DM, Held PJ, Merion RM. Risk Factors for End-Stage Renal Disease (ESRD) in Liver Transplant Recipients. Am J Transplant 2002, 2(Suppl 3):A450.
22. Wynn JJ, Port FK, Wolfe RA, Bragg JL, Rush SH, Ashby VB, Distant DA, Roberts JP, Held PJ. The Effect of HLA B and DR Points on Access to Kidney Transplantation and Graft Failure by Race and Ethnicity in the US. Am J Transplant 2002, 2(Suppl 3):A33.
1. Kayler LK, Rasmussen CS, Dykstra DM, Ojo AO, Port FK, Wolfe RA, Merion, RM. Gender Imbalance and Outcomes in Living Donor Renal Transplantation in the United States. Am J Transplant. In press.
2. Merion RM, Wolfe RA, Dykstra DM, Leichtman AB, Gillespie BW, Held PJ. Longitudinal Assessment of Mortality Risk Among Candidates for Liver Transplantation. Liver Transplantation. In press.
3. Port FK, Bragg JL, Metzger RA, Dykstra DM, Gillespie BW, Young EW, Delmonico FL, Wynn JJ, Merion RM, Wolfe RA, Held PJ. Donor Characteristics Associated With Reduced Graft Survival: an Approach to Expanding the Pool of Kidney Donors. Transplantation 2002, 74(9): 1281-1286.
Center-Specific Reports | OPO Reports | National Reports | Data Accuracy | Report Timeline
Home | Who We Are | What We Do | About Transplants | Transplant Statistics | Research Resources | Contact Us | Site Map
SRTR Site designed and maintained by Diamond Bullet Design & URREA.